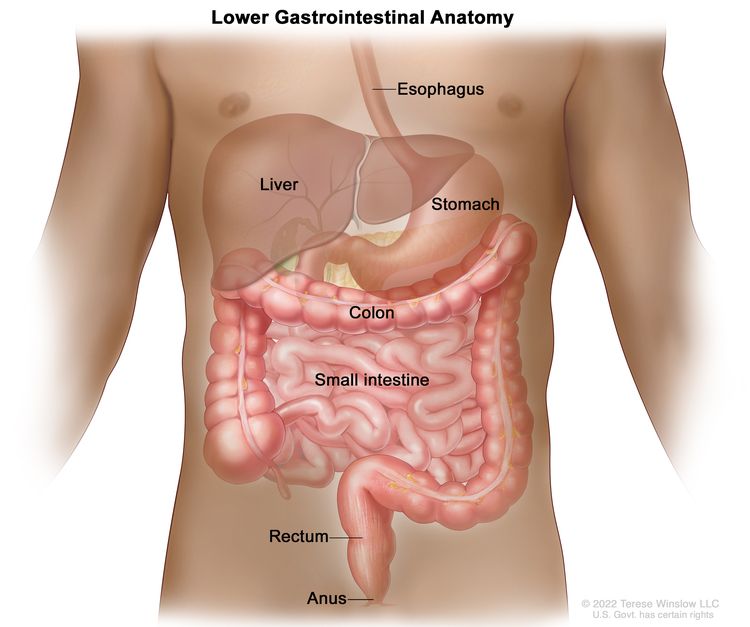
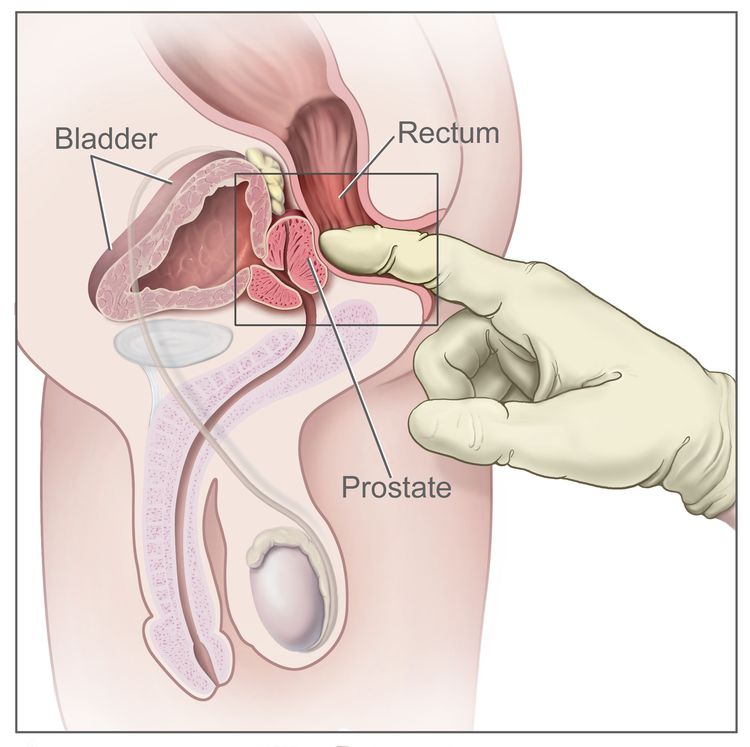
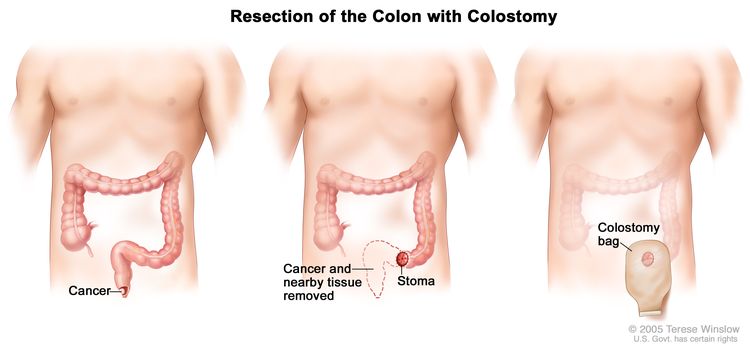
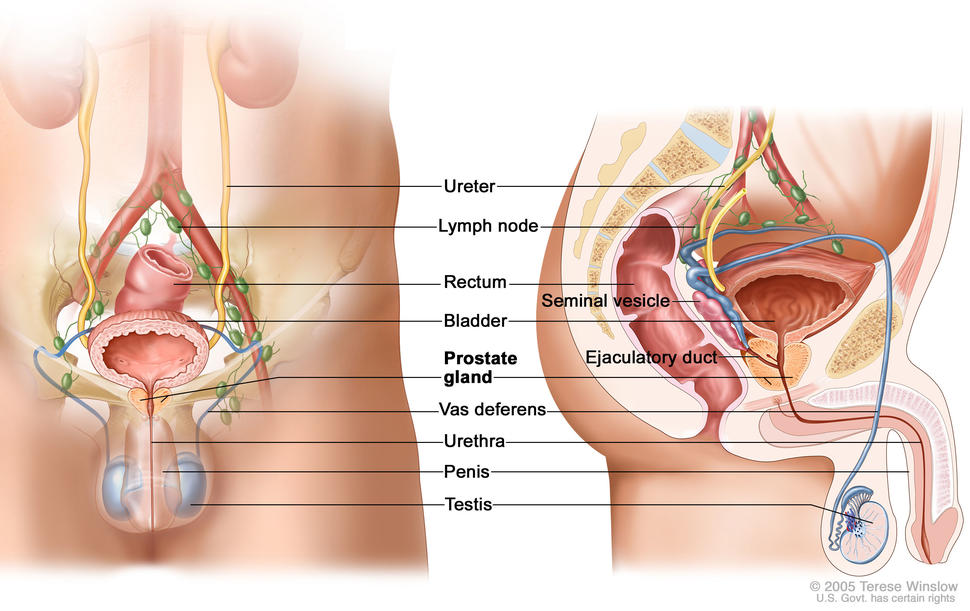
Standard treatments for prostate cancer that has not spread elsewhere in the body are surgery or radiation therapy, with or without hormone therapy.
Active surveillance is also an option for men who have a low risk of their cancer spreading. This means monitoring the cancer with regular biopsies and other tests, and holding off on treatment unless there is evidence of progression. Rates of active surveillance more than doubled between 2014 and 2021, to almost 60% of US men diagnosed with low-risk prostate cancer.
Hormone therapy for prostate cancer
Over the last decade, several new approaches to hormone therapy for advanced or metastatic prostate cancer have been approved for clinical use.
Many prostate cancers that originally respond to treatment with standard hormone therapy become resistant over time, resulting in castrate-resistant prostate cancer (CRPC). Four newer drugs have been shown to extend survival in some groups of men with CRPC. All inhibit the action of hormones that drive CRPC:
These drugs are now also used in some people whose prostate cancer still responds to standard hormone therapies but has spread elsewhere in the body (metastasized).
Scientists are continuing to study novel treatments and drugs, along with new combinations of existing treatments, in men with metastatic and castrate-resistant prostate cancer.
Hormone therapy for biochemically recurrent prostate cancer
A biochemical recurrence is a rise in the blood level of PSA in people with prostate cancer after treatment with surgery or radiation. In 2023, the FDA approved enzalutamide, given alone or with another drug called leuprolide, for some men who have a biochemical recurrence and are at high risk of their cancer spreading but don’t have signs on regular imaging that their cancer has come back.
Use of this drug combination can improve how long these men live without their cancer spreading. But it’s not yet known if using the drugs in this manner improves how long people live overall. Researchers are trying to determine which patients will benefit most from these types of treatments.
PARP inhibitors for prostate cancer
A PARP inhibitor is a substance that blocks an enzyme in cells called PARP. PARP helps repair DNA when it becomes damaged. Some prostate tumors have genetic changes that limit their ability to repair DNA damage. These tumors may be sensitive to treatment with PARP inhibitors. Some people also inherit genetic factors that limit their body’s ability to repair DNA damage. Prostate tumors in such people can also be treated with PARP inhibitors.
Two PARP inhibitors, olaparib (Lynparza) and rucaparib (Rubraca), have been approved for use alone in some men whose prostate cancer has such genetic changes and has metastasized, and whose disease has stopped responding to standard hormone treatments alone.
Ongoing studies are looking at combining PARP inhibitors with hormone therapies. Since 2023, the FDA has approved three such combinations for some men with metastatic prostate cancer:
Immunotherapy: vaccines for prostate cancer
Immunotherapies are treatments that harness the power of the immune system to fight cancer. These treatments can either help the immune system attack the cancer directly or stimulate the immune system in a more general way.
Vaccines and checkpoint inhibitors are two types of immunotherapy being tested in prostate cancer. Treatment vaccines are injections that stimulate the immune system to recognize and attack a tumor.
One type of treatment vaccine called sipuleucel-T (Provenge) is approved for men with few or no symptoms from metastatic CRPC.
Immunotherapy: checkpoint inhibitors for prostate cancer
An immune checkpoint inhibitor is a type of drug that blocks proteins on immune cells, making the immune system more effective at killing cancer cells.
Two checkpoint inhibitors, pembrolizumab (Keytruda) and dostarlimab (Jemperli) have been approved for the treatment of tumors, including prostate cancers, that have specific genetic features. Pembrolizumab has also been approved for any tumor that has metastasized and has a high number of genetic mutations.
But relatively few prostate cancers have these features, and prostate cancer in general has largely been resistant to treatment with checkpoint inhibitors and other immunotherapies, such as CAR T-cell therapy.
Research is ongoing to find ways to help the immune system recognize prostate tumors and help immune cells penetrate prostate tumor tissue. Studies are looking at whether combinations of immunotherapy drugs, or immunotherapy drugs given with other types of treatment, may be more effective in treating prostate cancer than single immunotherapies alone.
PSMA-targeted radiation therapy
Scientists have developed targeted therapies based on PSMA, the same protein that is used for imaging prostate cancer. For treatment, the molecule that targets PSMA is chemically linked to a radioactive substance. This new compound can potentially find, bind to, and kill prostate cancer cells throughout the body.
In a recent clinical trial, men with a type of advanced prostate cancer who received a PSMA-targeting drug lived longer than those who received standard therapies. This trial led to FDA approval of the drug, Lu177-PSMA-617 (Pluvicto), to treat some people with metastatic prostate cancer who had previously received chemotherapy.
An ongoing study is testing the use of Lu177-PSMA-617 in some people with metastatic prostate cancer who haven’t yet received chemotherapy. Other clinical trials are testing PSMA-targeting drugs in patients with earlier stages of prostate cancer, and in combination with other treatments, including targeted therapies like PARP inhibitors and immunotherapy.
Personalized clinical trials for prostate cancer
Research is uncovering more information about the genetic changes that happen as prostate cancers develop and progress. Although early-stage prostate cancer has relatively few genetic changes compared with other types of cancer, researchers have learned that metastatic prostate cancers usually accumulate more changes as they spread through the body.
These changes may make men with metastatic prostate cancers candidates for what are called “basket” clinical trials of new drugs. Such trials enroll participants based on the changes found in their cancer, not where in the body the cancer arose. In the NCI-MATCH trial, a high percentage of enrolled men with advanced prostate cancer had genetic changes that could potentially be targeted with investigational drugs.