Cancer of Unknown Primary (CUP) Treatment (PDQ®)–Health Professional Version
General Information About Cancer of Unknown Primary
A cancer of unknown primary (CUP) is defined as a biopsy-confirmed malignancy with no established primary site after pathological evaluation and radiographic studies. With the advent of comprehensive genomic profiling and positron emission tomography–computed tomography scanning, less than 1% of new cancer diagnoses are designated as CUP.[1–5]
Most CUPs are adenocarcinomas or undifferentiated tumors. Less commonly, squamous cell carcinomas, melanomas, sarcomas, germ cell tumors, and neuroendocrine tumors present with an undetermined primary site of origin.[6] In some patients, the primary site cannot be identified even at postmortem examination.
Prognosis and Survival
The prognosis for patients with CUP is poor. Approximately 30% of patients are alive at 1 year.[1,7] CUP is represented by a heterogeneous group of diseases, all of which have presented with metastasis as the primary manifestation. Although most diseases are relatively refractory to cytotoxic treatments, molecular and genetic diagnostics can identify targeted therapies that may result in a much better prognosis.[1] In each instance, the clinician must consider distinct clinical and pathological details when selecting appropriate, and potentially curative, management.
Survival is worse for patients with any of the following characteristics: [8–10]
- Adenocarcinoma or undifferentiated carcinoma histology.
- Extranodal presentation.
- Older age.
- Hepatic or adrenal involvement.
- Poor performance status.
- Elevated serum lactate dehydrogenase (LDH).
Lymph node involvement or neuroendocrine histology were associated with longer survival in retrospective reviews.[8–10]
Patients who present with LDH levels within the reference range and an Eastern Cooperative Oncology Group performance status of 0 or 1 have better prognoses than those with high LDH levels or worse performance statuses.[10]
A review of five case control studies and 14 cohort studies found a clear increased risk of CUP with smoking.[11]
Patterns of Metastases
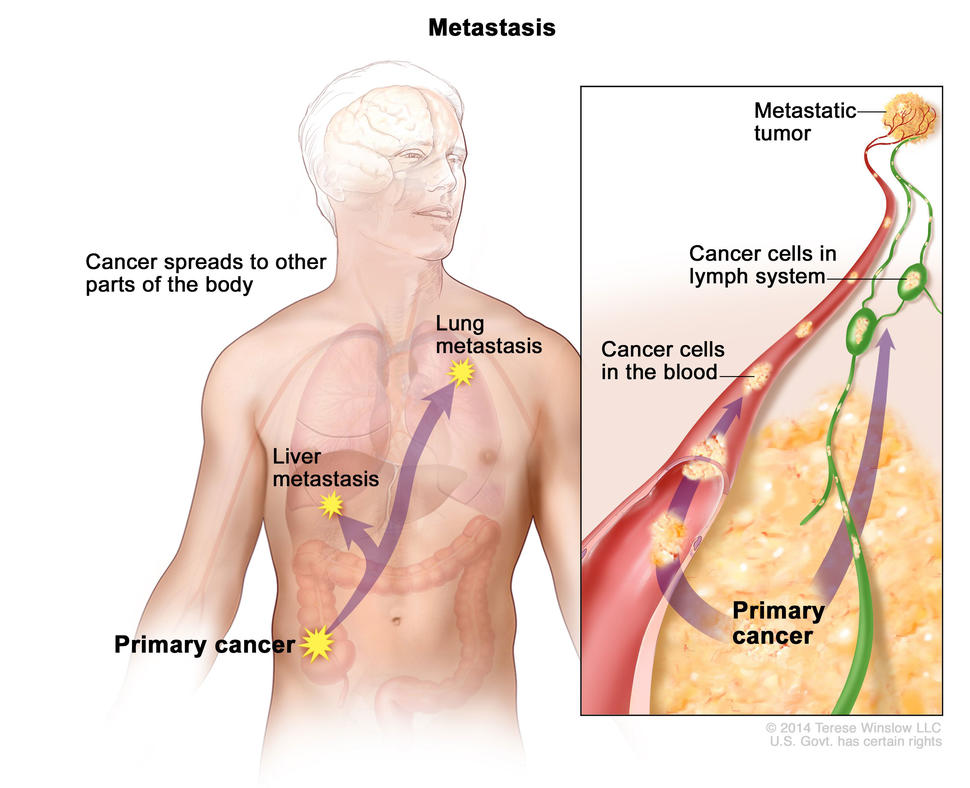
Conceptually, CUP is a type of tumor that tends to metastasize early, unlike more common cases, in which the primary tumor is apparent, with or without metastasis. The pattern of spread of CUP at diagnosis can provide clues to the likelihood of the primary site being above or below the diaphragm. Lung metastases are twice as common in primary sites ultimately found to be above the diaphragm. Liver metastases are more common from primary disease sites below the diaphragm. A cancer presenting as CUP may have a significantly different pattern of metastasis than what is typically expected. For instance, when presenting as CUP, bone metastases are more common from pancreatic cancer and less common from lung cancer, unlike their typical presentation.
Diagnostic Evaluation
The pathologist has a central role in the evaluation of CUP. A thorough evaluation of an adequate specimen using histology, immunohistochemistry, molecular diagnostics, next-generation sequencing (NGS), and, when appropriate, electron microscopy provides the most important clues in the diagnosis of CUP.[12–16] Pathological evaluations provide guidance for an appropriate clinical evaluation.
The following tests may be used to diagnose CUP:
- Gene expression profiling: Gene expression profiling and NGS may identify a potential site of origin in patients with CUP.[5,12–14,17]
- Beta-human chorionic gonadotropin (beta-hCG) and alpha-fetoprotein (AFP) analysis: Beta-hCG and AFP are not absolutely specific for germ cell tumors, and AFP is not specific for hepatoma. However, abnormal levels of these proteins are an important finding in young men with a mediastinal or retroperitoneal mass. Prognosis is improved for patients with germ cell tumors treated with site-specific therapy.[18]
- Other tumor marker tests include prostate-specific antigen (prostate cancer) in men; CA-125 (ovarian or gynecologic cancers), CA 15-3 (breast cancer), and CA 27.29 (breast cancer) in women; and carcinoembryonic antigen (CEA), CA 19-9, CA 72-4 (adenocarcinoma), and chromogranin A (neuroendocrine cancer). Elevation of less specific tumor-specific markers such as CEA, CA 19-9, and CA 72-4 can be used to monitor progression or tumor response.
- Magnetic resonance imaging is used for head and neck presentations, brain metastases, suspected pelvic neoplasms, suspected breast cancer (with isolated axillary lymph nodes or isolated bone metastases), prostate lesions, and adrenal lesions.
- For patients with presumed CUP, a prior diagnosis of malignancy should be evaluated by pathological comparison, using genomic sequencing in both tissue and circulating tumor DNA, when available.[19]
Clinical, pathological, and molecular genetic tests have two goals. The first is to identify tumors that are responsive to available therapies. The second is to identify novel targeted therapies that might be applicable to the particular genetic profile.[5] The U.S. Food and Drug Administration (FDA) has approved some novel targeted therapies when a molecular target is identified in a cancer, regardless of primary site (including when no primary site is evident with metastases). Examples include tumors with the following actionable targets:[20–27]
- High tumor mutational burden.
- Elevated PD-1 or PD-L1 expression.
- HER2 expression.
- Pathogenic variants in NTRK, RET, or EGFR.
- BRAF V600E pathogenic variant.
- ROS1 gene fusion.
- MET amplification.
- Homologous recombination repair deficiency/microsatellite instability.
Two randomized prospective trials published in 2019 compared standard chemotherapy (carboplatin and paclitaxel or cisplatin and gemcitabine) versus site-specific therapy using gene expression profiling.[28,29] There was no advantage to site-specific therapy in median progression-free survival (PFS) (5 months) or median overall survival (OS) (10–12 months) in either trial. However, most novel targeted therapies, including checkpoint inhibitors, were not available at that time.[28,29][Level of evidence B1]
A meta-analysis of five studies including 1,114 patients compared site-specific therapy from genomic profiling versus empiric therapy. There was no improvement in PFS (hazard ratio [HR], 0.93; 95% confidence interval [CI], 0.74–1.17; P = .534) or OS (HR, 0.75; 95% CI, 0.55–1.03; P = .069).[30][Level of evidence B1] Isolated benefit was seen in patients with more responsive tumors with an identified specifically targeted therapy.
In a retrospective analysis, a cohort of 97 patients with CUP underwent NGS of tissue- or blood-derived cell-free DNA (cfDNA). The median number of pathogenic genomic alterations found in tissue was four (range, 0–25), and the median number of genomic alterations found in cfDNA was two (range, 0–9).[31] Combination matched therapies based on these alterations might improve patient outcomes.[31] A matching score (MS) (roughly equivalent to the number of alterations targeted/total number of deleterious alterations) was calculated post hoc to quantify the degree of the tumor-to-drug match. Outcomes were compared for evaluable patients determined to be MS high (>50%; n = 15) or MS low (≤50%; n = 47). The median PFS was 10.4 months for MS-high patients and 2.8 months for MS-low patients (HR, 0.27; 95% CI, 0.11–0.64; P = .002). The OS was 15.8 months for MS-high patients and 6.9 months for MS-low patients (HR, 0.45; 95% CI, 0.17–1.16; P = .09).[31][Level of evidence C2]
References
- Binder C, Matthes KL, Korol D, et al.: Cancer of unknown primary-Epidemiological trends and relevance of comprehensive genomic profiling. Cancer Med 7 (9): 4814-4824, 2018. [PUBMED Abstract]
- Kwee TC, Kwee RM: Combined FDG-PET/CT for the detection of unknown primary tumors: systematic review and meta-analysis. Eur Radiol 19 (3): 731-44, 2009. [PUBMED Abstract]
- Burglin SA, Hess S, Høilund-Carlsen PF, et al.: 18F-FDG PET/CT for detection of the primary tumor in adults with extracervical metastases from cancer of unknown primary: A systematic review and meta-analysis. Medicine (Baltimore) 96 (16): e6713, 2017. [PUBMED Abstract]
- Woo S, Becker AS, Do RKG, et al.: Impact of 18F-Fluorodeoxyglucose positron emission tomography on management of cancer of unknown primary: systematic review and meta-analysis. Eur J Cancer 159: 60-77, 2021. [PUBMED Abstract]
- Conway AM, Mitchell C, Kilgour E, et al.: Molecular characterisation and liquid biomarkers in Carcinoma of Unknown Primary (CUP): taking the ‘U’ out of ‘CUP’. Br J Cancer 120 (2): 141-153, 2019. [PUBMED Abstract]
- Krämer A, Bochtler T, Pauli C, et al.: Cancer of unknown primary: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol 34 (3): 228-246, 2023. [PUBMED Abstract]
- Varadhachary GR, Raber MN: Cancer of unknown primary site. N Engl J Med 371 (8): 757-65, 2014. [PUBMED Abstract]
- Hess KR, Abbruzzese MC, Lenzi R, et al.: Classification and regression tree analysis of 1000 consecutive patients with unknown primary carcinoma. Clin Cancer Res 5 (11): 3403-10, 1999. [PUBMED Abstract]
- Hemminki K, Bevier M, Hemminki A, et al.: Survival in cancer of unknown primary site: population-based analysis by site and histology. Ann Oncol 23 (7): 1854-63, 2012. [PUBMED Abstract]
- Culine S, Kramar A, Saghatchian M, et al.: Development and validation of a prognostic model to predict the length of survival in patients with carcinomas of an unknown primary site. J Clin Oncol 20 (24): 4679-83, 2002. [PUBMED Abstract]
- Hermans KEPE, Kazemzadeh F, Loef C, et al.: Risk factors for cancer of unknown primary: a literature review. BMC Cancer 23 (1): 314, 2023. [PUBMED Abstract]
- Horlings HM, van Laar RK, Kerst JM, et al.: Gene expression profiling to identify the histogenetic origin of metastatic adenocarcinomas of unknown primary. J Clin Oncol 26 (27): 4435-41, 2008. [PUBMED Abstract]
- Handorf CR, Kulkarni A, Grenert JP, et al.: A multicenter study directly comparing the diagnostic accuracy of gene expression profiling and immunohistochemistry for primary site identification in metastatic tumors. Am J Surg Pathol 37 (7): 1067-75, 2013. [PUBMED Abstract]
- Ross JS, Wang K, Gay L, et al.: Comprehensive Genomic Profiling of Carcinoma of Unknown Primary Site: New Routes to Targeted Therapies. JAMA Oncol 1 (1): 40-9, 2015. [PUBMED Abstract]
- Selves J, Long-Mira E, Mathieu MC, et al.: Immunohistochemistry for Diagnosis of Metastatic Carcinomas of Unknown Primary Site. Cancers (Basel) 10 (4): , 2018. [PUBMED Abstract]
- Pauli C, Bochtler T, Mileshkin L, et al.: A Challenging Task: Identifying Patients with Cancer of Unknown Primary (CUP) According to ESMO Guidelines: The CUPISCO Trial Experience. Oncologist 26 (5): e769-e779, 2021. [PUBMED Abstract]
- Ross JS, Sokol ES, Moch H, et al.: Comprehensive Genomic Profiling of Carcinoma of Unknown Primary Origin: Retrospective Molecular Classification Considering the CUPISCO Study Design. Oncologist 26 (3): e394-e402, 2021. [PUBMED Abstract]
- Motzer RJ, Rodriguez E, Reuter VE, et al.: Molecular and cytogenetic studies in the diagnosis of patients with poorly differentiated carcinomas of unknown primary site. J Clin Oncol 13 (1): 274-82, 1995. [PUBMED Abstract]
- Bochtler T, Endris V, Leichsenring J, et al.: Comparative genetic profiling aids diagnosis and clinical decision making in challenging cases of CUP syndrome. Int J Cancer 145 (11): 2963-2973, 2019. [PUBMED Abstract]
- Adashek JJ, Menta AK, Reddy NK, et al.: Tissue-Agnostic Activity of BRAF plus MEK Inhibitor in BRAF V600-Mutant Tumors. Mol Cancer Ther 21 (6): 871-878, 2022. [PUBMED Abstract]
- Subbiah V, Wolf J, Konda B, et al.: Tumour-agnostic efficacy and safety of selpercatinib in patients with RET fusion-positive solid tumours other than lung or thyroid tumours (LIBRETTO-001): a phase 1/2, open-label, basket trial. Lancet Oncol 23 (10): 1261-1273, 2022. [PUBMED Abstract]
- Subbiah V, Cassier PA, Siena S, et al.: Pan-cancer efficacy of pralsetinib in patients with RET fusion-positive solid tumors from the phase 1/2 ARROW trial. Nat Med 28 (8): 1640-1645, 2022. [PUBMED Abstract]
- Le DT, Uram JN, Wang H, et al.: PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N Engl J Med 372 (26): 2509-20, 2015. [PUBMED Abstract]
- Overman MJ, McDermott R, Leach JL, et al.: Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): an open-label, multicentre, phase 2 study. Lancet Oncol 18 (9): 1182-1191, 2017. [PUBMED Abstract]
- Luchini C, Bibeau F, Ligtenberg MJL, et al.: ESMO recommendations on microsatellite instability testing for immunotherapy in cancer, and its relationship with PD-1/PD-L1 expression and tumour mutational burden: a systematic review-based approach. Ann Oncol 30 (8): 1232-1243, 2019. [PUBMED Abstract]
- Marcus L, Lemery SJ, Keegan P, et al.: FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors. Clin Cancer Res 25 (13): 3753-3758, 2019. [PUBMED Abstract]
- Tanizaki J, Yonemori K, Akiyoshi K, et al.: Open-label phase II study of the efficacy of nivolumab for cancer of unknown primary. Ann Oncol 33 (2): 216-226, 2022. [PUBMED Abstract]
- Fizazi K, Maillard A, Penel N: A phase III trial of empiric chemotherapy with cisplatin and gemcitabine or systemic treatment tailored by molecular gene expression analysis in patients with carcinomas of an unknown primary (CUP) site (GEFCAPI 04). [Abstract] Ann Oncol 30 (Suppl 5): A-LBA15_PR, v851, 2019.
- Hayashi H, Kurata T, Takiguchi Y, et al.: Randomized Phase II Trial Comparing Site-Specific Treatment Based on Gene Expression Profiling With Carboplatin and Paclitaxel for Patients With Cancer of Unknown Primary Site. J Clin Oncol 37 (7): 570-579, 2019. [PUBMED Abstract]
- Ding Y, Jiang J, Xu J, et al.: Site-specific therapy in cancers of unknown primary site: a systematic review and meta-analysis. ESMO Open 7 (2): 100407, 2022. [PUBMED Abstract]
- Kato S, Gumas S, Adashek JJ, et al.: Multi-omic analysis in carcinoma of unknown primary (CUP): therapeutic impact of knowing the unknown. Mol Oncol 18 (4): 956-968, 2024. [PUBMED Abstract]
Stage Information for Cancer of Unknown Primary
A positron emission tomography–computed tomography (PET-CT) scan of the whole body can reveal the extent of disease and is especially useful in single-site or oligometastatic presentations, especially for patients with cervical lymph node metastases suspicious for a head and neck primary.[1–4]
PET-CT can be combined with somatostatin receptor PET-CT scans for neuroendocrine neoplasms or prostate-specific membrane antigen for suspected prostate cancer. A meta-analysis of 20 studies including 1,942 patients found a biopsy confirmed or clinically confirmed positive PET-CT scan in 40.9% of patients (95% confidence interval [CI], 40.0%–42.9%).[5]
A meta-analysis of 38 studies including 2,795 patients found that a fluorine F 18-fludeoxyglucose–PET or PET-CT scan led to management changes in 35% of patients (95% CI, 31%–40%).[6] In this meta-analysis, PET-CT identified a primary site in 22% of patients (95% CI, 18%–28%) and other metastases in 14% of patients (95% CI, 10%–19%).[6]
After staging with a PET-CT scan and magnetic resonance imaging of the head, the next step is molecular characterization and next-generation sequencing, preferably on the tissue biopsy or alternatively by analyzing circulating tumor cells on a blood sample (so-called liquid biopsy).
References
- Lievens Y, Guckenberger M, Gomez D, et al.: Defining oligometastatic disease from a radiation oncology perspective: An ESTRO-ASTRO consensus document. Radiother Oncol 148: 157-166, 2020. [PUBMED Abstract]
- Albertson M, Chandra S, Sayed Z, et al.: PET/CT Evaluation of Head and Neck Cancer of Unknown Primary. Semin Ultrasound CT MR 40 (5): 414-423, 2019. [PUBMED Abstract]
- de Braud F, al-Sarraf M: Diagnosis and management of squamous cell carcinoma of unknown primary tumor site of the neck. Semin Oncol 20 (3): 273-8, 1993. [PUBMED Abstract]
- Smith KA, Dort JC, Hall SF, et al.: Cost-effectiveness of positron emission tomography-CT in the evaluation of cancer of unknown primary of the head and neck. Head Neck 37 (12): 1781-7, 2015. [PUBMED Abstract]
- Burglin SA, Hess S, Høilund-Carlsen PF, et al.: 18F-FDG PET/CT for detection of the primary tumor in adults with extracervical metastases from cancer of unknown primary: A systematic review and meta-analysis. Medicine (Baltimore) 96 (16): e6713, 2017. [PUBMED Abstract]
- Woo S, Becker AS, Do RKG, et al.: Impact of 18F-Fluorodeoxyglucose positron emission tomography on management of cancer of unknown primary: systematic review and meta-analysis. Eur J Cancer 159: 60-77, 2021. [PUBMED Abstract]
Treatment of Newly Diagnosed Favorable Cancer of Unknown Primary
Twenty percent of patients with cancer of unknown primary (CUP) have favorable CUP. Favorable CUP is defined by the following characteristics:
- Single-site or oligometastatic disease amenable to surgery or radiation therapy.
- Cervical lymph node(s) with squamous cell histology (head and neck paradigm).
- Inguinal lymph nodes with squamous cell histology (genitourinary or anorectal source).
- Axillary lymph node(s) with adenocarcinoma histology (breast paradigm).
- Bone metastases if blastic (breast paradigm for women, prostate paradigm for men).
- Elevated alpha-fetoprotein (AFP) and/or beta-human chorionic gonadotropin (beta-hCG) (germ cell paradigm).
- Peritoneal carcinomatosis if adenocarcinoma (ovarian paradigm).
- Targeted therapy defined by molecular markers or by next-generation sequencing (including PD-L1, homologous recombination repair deficiency [HRD], microsatellite instability [MSI]).
- Neuroendocrine neoplasms.
- Melanoma (melanotic or amelanotic) occurring in a single lymph node site.
Single-Site or Oligometastatic Disease Amenable to Surgery or Radiation Therapy
According to a consensus paper, when a single site of metastasis can be confirmed by positron emission tomography–computed tomography (PET-CT) scan and magnetic resonance imaging (MRI) of the brain, local treatment with surgery or radiation therapy is an option. Local treatment has resulted in anecdotal cure in a few patients and several months to years of anecdotal relapse-free survival in a few patients.[1][Level of evidence C3] Anecdotal reports also support localized therapy for patients with fewer than five metastases, or even re-treatment with radiation therapy or surgery for patients with localized recurrence.[2][Level of evidence C3] A patient staged with CUP and a single brain metastasis may also do well with radiation therapy with or without surgery, as was reported in a prospective study of 33 patients.[3,4][Level of evidence C3]
Cervical Lymph Node(s) With Squamous Cell Histology (Head and Neck Paradigm)
PET-CT, MRI, and pan-endoscopy for biopsies and bilateral tonsillectomies are performed when squamous cell carcinoma is identified in cervical lymph nodes (not supraclavicular) in patients with CUP.[5]
Transoral robotic surgery may replace pan-endoscopy in the search for a primary site in the head and neck region.[6] Pathological studies should include testing for p16 expression to assess human papillomavirus status, Epstein-Barr virus status, and PD-L1 expression.[7]
For more information, see Metastatic Squamous Neck Cancer with Occult Primary Treatment.
Inguinal Lymph Nodes With Squamous Cell Histology (Genitourinary or Anorectal Source)
Squamous cell carcinoma in an inguinal or iliac site is usually associated with a genital or anorectal primary. Examinations should focus on the vulva, vagina, and cervix in women, and the penis in men. In both sexes, the anorectal area should be examined, and any suspicious areas should be biopsied. Anecdotal cures have been reported in a few cases of isolated inguinal or iliac sites with an occult primary after staging studies.[8]
Axillary Lymph Node(s) With Adenocarcinoma Histology (Breast Paradigm)
Less than 1% of female patients present with histological breast cancer in an axillary lymph node with an occult primary even after MRI of the breast.[9] Initial pathological studies should include estrogen-receptor, progesterone-receptor, and human epidermal growth factor (HER2) status, with further molecular and genetic studies done subsequently. Surgery, radiation therapy, neoadjuvant or adjuvant chemotherapy, and/or hormonal therapy are given according to guidelines for conventional breast cancer.[10–13]
For more information, see Breast Cancer Treatment.
Bone Metastases if Blastic (Breast Paradigm for Women, Prostate Paradigm for Men)
The presence of blastic bone metastases or mixed blastic/lytic lesions will usually suggest breast cancer in women and prostate cancer in men. Diagnostic procedures and therapeutic options should proceed in these directions.
For more information, see Breast Cancer Treatment or Prostate Cancer Treatment.
Elevated AFP and/or Beta-hCG (Germ Cell Paradigm)
The presence of elevated serum or tissue levels of AFP and/or beta-hCG suggests testicular cancer or extragonadal germ cell tumor in males. Men younger than 50 years with mediastinal adenopathy and pulmonary metastases have immunohistochemical evidence of AFP and/or beta-hCG in the extragonadal presentation. Diagnostic procedures and therapeutic options should proceed in these directions.
For more information, see Testicular Cancer Treatment and Extragonadal Germ Cell Tumors Treatment.
Peritoneal Carcinomatosis if Adenocarcinoma (Ovarian Paradigm)
Women with peritoneal carcinomatosis of an undifferentiated adenocarcinoma or serous histology in the absence of an ovarian, fallopian tube, or uterine primary site should undergo diagnostic testing and a therapeutic approach for ovarian cancer.[14] This is considered a primary peritoneal serous carcinoma.
For more information, see Ovarian Epithelial, Fallopian Tube, and Primary Peritoneal Cancer Treatment.
Targeted Therapy Defined by Molecular Markers or by Next-Generation Sequencing (Including PD-L1, HRD, MSI)
Clinical, pathological, and molecular genetic tests have two goals. The first is to identify tumors that are responsive to available therapies. The second is to identify novel targeted therapies that might be applicable to the particular genetic profile. The U.S. Food and Drug Administration has approved some novel targeted therapies when a molecular target is identified in a cancer, regardless of primary site or when no primary site is evident. Examples include tumors with the following actionable targets:[15–22]
- High tumor mutational burden.
- Elevated PD-1 or PD-L1 expression.
- HER2 expression.
- Pathogenic variants in NTRK, RET, or EGFR.
- BRAF V600E pathogenic variant.
- ROS1 gene fusion.
- MET amplification.
- HRD/MSI.
Neuroendocrine Neoplasms
Neuroendocrine neoplasms are classified as well-differentiated neuroendocrine tumors (NETs) and slow progression neuroendocrine carcinomas (NEC). Well-differentiated NETs exhibit relatively indolent behavior. Slow progression NECs may exhibit highly aggressive behavior, with a rapid metastatic spread that is clinically indistinguishable from pancreatic adenocarcinoma or small cell lung cancer. The Ki-67 proliferative index rate is used to subclassify well-differentiated NETs into low, intermediate, and high-grade, while NECs are by default high-grade and poorly differentiated and divided into small- and large-cell NEC.
NETs of unknown primary origin may derive from the small bowel, pancreas, appendix, colon, rectum, or ovary.[23,24] Somatostatin receptor PET-CT scans are highly effective at identifying primary sites and metastatic sites for well-differentiated NETs, while fluorine F 18-fludeoxyglucose–PET is more appropriate for NEC.[25–27]
For more information, see Gastrointestinal Neuroendocrine Tumors Treatment.
Merkel cell carcinoma is a rare, aggressive, cutaneous malignancy of neuroendocrine origin, usually with primary lesions in the head and neck or in the extremities. However, it may present in the lymph nodes only, with no evident primary site.[28]
For more information, see Merkel Cell Carcinoma Treatment.
Melanoma (Melanotic or Amelanotic) Occurring in a Single Lymph Node Site
Approximately 5% of patients with malignant melanoma will present without a documented primary site. Diagnostic studies and therapeutic options should proceed with the paradigm of malignant melanoma.[29]
For more information, see Melanoma Treatment.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Lievens Y, Guckenberger M, Gomez D, et al.: Defining oligometastatic disease from a radiation oncology perspective: An ESTRO-ASTRO consensus document. Radiother Oncol 148: 157-166, 2020. [PUBMED Abstract]
- Pouyiourou M, Wohlfromm T, Kraft B, et al.: Local ablative treatment with surgery and/or radiotherapy in single-site and oligometastatic carcinoma of unknown primary. Eur J Cancer 157: 179-189, 2021. [PUBMED Abstract]
- Bartelt S, Lutterbach J: Brain metastases in patients with cancer of unknown primary. J Neurooncol 64 (3): 249-53, 2003. [PUBMED Abstract]
- Rudà R, Borgognone M, Benech F, et al.: Brain metastases from unknown primary tumour: a prospective study. J Neurol 248 (5): 394-8, 2001. [PUBMED Abstract]
- de Braud F, al-Sarraf M: Diagnosis and management of squamous cell carcinoma of unknown primary tumor site of the neck. Semin Oncol 20 (3): 273-8, 1993. [PUBMED Abstract]
- Al-Lami A, Gao C, Saddiq M, et al.: Reducing the unknowns: A systematic review & meta-analysis of the effectiveness of trans-oral surgical techniques in identifying head and neck primary cancer in carcinoma unknown primary. Oral Oncol 126: 105748, 2022. [PUBMED Abstract]
- Machiels JP, René Leemans C, Golusinski W, et al.: Squamous cell carcinoma of the oral cavity, larynx, oropharynx and hypopharynx: EHNS-ESMO-ESTRO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 31 (11): 1462-1475, 2020. [PUBMED Abstract]
- Matsuyama S, Nakafusa Y, Tanaka M, et al.: Iliac lymph node metastasis of an unknown primary tumor: report of a case. Surg Today 36 (7): 655-8, 2006. [PUBMED Abstract]
- de Bresser J, de Vos B, van der Ent F, et al.: Breast MRI in clinically and mammographically occult breast cancer presenting with an axillary metastasis: a systematic review. Eur J Surg Oncol 36 (2): 114-9, 2010. [PUBMED Abstract]
- Kim H, Park W, Kim SS, et al.: Prognosis of patients with axillary lymph node metastases from occult breast cancer: analysis of multicenter data. Radiat Oncol J 39 (2): 107-112, 2021. [PUBMED Abstract]
- McCartan DP, Zabor EC, Morrow M, et al.: Oncologic Outcomes After Treatment for MRI Occult Breast Cancer (pT0N+). Ann Surg Oncol 24 (11): 3141-3147, 2017. [PUBMED Abstract]
- Van den Bruele AB, Chen I, Sevilimedu V, et al.: Management of ipsilateral breast tumor recurrence following breast conservation surgery: a comparative study of re-conservation vs mastectomy. Breast Cancer Res Treat 187 (1): 105-112, 2021. [PUBMED Abstract]
- Thompson JL, Sinco BR, McCaffrey RL, et al.: Prophylactic mastectomy and occult malignancy: Surgical and imaging considerations. J Surg Oncol 127 (1): 18-27, 2023. [PUBMED Abstract]
- Pentheroudakis G, Pavlidis N: Serous papillary peritoneal carcinoma: unknown primary tumour, ovarian cancer counterpart or a distinct entity? A systematic review. Crit Rev Oncol Hematol 75 (1): 27-42, 2010. [PUBMED Abstract]
- Adashek JJ, Menta AK, Reddy NK, et al.: Tissue-Agnostic Activity of BRAF plus MEK Inhibitor in BRAF V600-Mutant Tumors. Mol Cancer Ther 21 (6): 871-878, 2022. [PUBMED Abstract]
- Subbiah V, Wolf J, Konda B, et al.: Tumour-agnostic efficacy and safety of selpercatinib in patients with RET fusion-positive solid tumours other than lung or thyroid tumours (LIBRETTO-001): a phase 1/2, open-label, basket trial. Lancet Oncol 23 (10): 1261-1273, 2022. [PUBMED Abstract]
- Subbiah V, Cassier PA, Siena S, et al.: Pan-cancer efficacy of pralsetinib in patients with RET fusion-positive solid tumors from the phase 1/2 ARROW trial. Nat Med 28 (8): 1640-1645, 2022. [PUBMED Abstract]
- Le DT, Uram JN, Wang H, et al.: PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N Engl J Med 372 (26): 2509-20, 2015. [PUBMED Abstract]
- Overman MJ, McDermott R, Leach JL, et al.: Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): an open-label, multicentre, phase 2 study. Lancet Oncol 18 (9): 1182-1191, 2017. [PUBMED Abstract]
- Luchini C, Bibeau F, Ligtenberg MJL, et al.: ESMO recommendations on microsatellite instability testing for immunotherapy in cancer, and its relationship with PD-1/PD-L1 expression and tumour mutational burden: a systematic review-based approach. Ann Oncol 30 (8): 1232-1243, 2019. [PUBMED Abstract]
- Marcus L, Lemery SJ, Keegan P, et al.: FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors. Clin Cancer Res 25 (13): 3753-3758, 2019. [PUBMED Abstract]
- Tanizaki J, Yonemori K, Akiyoshi K, et al.: Open-label phase II study of the efficacy of nivolumab for cancer of unknown primary. Ann Oncol 33 (2): 216-226, 2022. [PUBMED Abstract]
- Kotteas EA, Pavlidis N: Neuroendocrine Merkel cell nodal carcinoma of unknown primary site: management and outcomes of a rare entity. Crit Rev Oncol Hematol 94 (1): 116-21, 2015. [PUBMED Abstract]
- Begum N, Wellner U, Thorns C, et al.: CUP Syndrome in Neuroendocrine Neoplasia: Analysis of Risk Factors and Impact of Surgical Intervention. World J Surg 39 (6): 1443-51, 2015. [PUBMED Abstract]
- Ma H, Kan Y, Yang JG: Clinical value of 68Ga-DOTA-SSTR PET/CT in the diagnosis and detection of neuroendocrine tumors of unknown primary origin: a systematic review and meta-analysis. Acta Radiol 62 (9): 1217-1228, 2021. [PUBMED Abstract]
- Menda Y, O’Dorisio TM, Howe JR, et al.: Localization of Unknown Primary Site with 68Ga-DOTATOC PET/CT in Patients with Metastatic Neuroendocrine Tumor. J Nucl Med 58 (7): 1054-1057, 2017. [PUBMED Abstract]
- De Dosso S, Treglia G, Pascale M, et al.: Detection rate of unknown primary tumour by using somatostatin receptor PET/CT in patients with metastatic neuroendocrine tumours: a meta-analysis. Endocrine 64 (3): 456-468, 2019. [PUBMED Abstract]
- Broida SE, Chen XT, Baum CL, et al.: Merkel cell carcinoma of unknown primary: Clinical presentation and outcomes. J Surg Oncol 126 (6): 1080-1086, 2022. [PUBMED Abstract]
- De Andrade JP, Wong P, O’Leary MP, et al.: Multidisciplinary Care for Melanoma of Unknown Primary: Experience in the Era of Molecular Profiling. Ann Surg Oncol 27 (13): 5240-5247, 2020. [PUBMED Abstract]
Treatment of Newly Diagnosed Unfavorable Cancer of Unknown Primary
Most patients with newly diagnosed cancer of unknown primary (CUP) are considered to have an unfavorable prognosis.
Treatment Options for Newly Diagnosed Unfavorable CUP
Treatment options for newly diagnosed unfavorable CUP include the following:
Site-directed therapy based on molecular tissue of origin or molecular targeted therapy
Clinical, pathological, and molecular genetic tests have two goals. The first is to identify tumors that are responsive to available therapies. The second is to identify novel targeted therapies that might be applicable to the particular genetic profile. The U.S. Food and Drug Administration (FDA) has approved some novel targeted therapies when a molecular target is identified in a cancer, regardless of primary site or when no primary site is evident. Examples include tumors with the following actionable targets:[1–8]
- High tumor mutational burden (TMB-H).
- Elevated PD-1 or PD-L1 expression.
- HER2 expression.
- Pathogenic variants in NTRK, RET, or EGFR.
- BRAF V600E pathogenic variant.
- ROS1 gene fusion.
- MET amplification.
- Homologous recombination repair deficiency/microsatellite instability (MSI).
Immunological therapy
Immunological therapy using checkpoint inhibitors such as nivolumab has been given to a general population with unfavorable CUP, resulting in a 22% overall response rate.[8] Better response rates can be seen when checkpoint inhibitors are given to patients with tumors expressing high levels of MSI (MSI-H) or deficient mismatch repair (dMMR).[7] Patients with TMB-H disease, defined as at least 7.75 mutations [8] or 10 mutations [9] per megabase, also have higher response rates to immunotherapy.[6] The FDA has approved pembrolizumab in a tumor-agnostic situation (which includes CUP) for tumors with MSI-H, dMMR, or TMB-H.[7] High PD-L1 expression also correlates with higher response rates in patients who receive immunotherapy.[8] Further trials for patients with CUP are required to assess when to give checkpoint inhibitors and to define optimal cutoffs (1% to 50%) and scoring systems (cancer cell tumor proportion score vs. cancer-plus-surrounding cell combined positive score) to guide treatment.[10]
Chemotherapy
Patients with unfavorable CUP who are not candidates for molecular targeted therapy or immunotherapy are candidates for clinical trials. Cytotoxic chemotherapy can be a palliative therapy, but the choice of drugs has been based on a few small clinical studies, with no randomized trials establishing their benefit over best supportive care.[10] Several randomized prospective studies demonstrate that platinum-based doublet chemotherapy combined with a taxane or gemcitabine is equivalent to other chemotherapy in response rate and progression-free survival.[11–14][Level of evidence B1] Although most patients in these trials had adenocarcinoma or poorly differentiated carcinoma, patients with poorly differentiated squamous cell carcinoma were included.
Palliative care and hospice are options for patients with a poor performance status and relapsing disease.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
References
- Adashek JJ, Menta AK, Reddy NK, et al.: Tissue-Agnostic Activity of BRAF plus MEK Inhibitor in BRAF V600-Mutant Tumors. Mol Cancer Ther 21 (6): 871-878, 2022. [PUBMED Abstract]
- Subbiah V, Wolf J, Konda B, et al.: Tumour-agnostic efficacy and safety of selpercatinib in patients with RET fusion-positive solid tumours other than lung or thyroid tumours (LIBRETTO-001): a phase 1/2, open-label, basket trial. Lancet Oncol 23 (10): 1261-1273, 2022. [PUBMED Abstract]
- Subbiah V, Cassier PA, Siena S, et al.: Pan-cancer efficacy of pralsetinib in patients with RET fusion-positive solid tumors from the phase 1/2 ARROW trial. Nat Med 28 (8): 1640-1645, 2022. [PUBMED Abstract]
- Le DT, Uram JN, Wang H, et al.: PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N Engl J Med 372 (26): 2509-20, 2015. [PUBMED Abstract]
- Overman MJ, McDermott R, Leach JL, et al.: Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): an open-label, multicentre, phase 2 study. Lancet Oncol 18 (9): 1182-1191, 2017. [PUBMED Abstract]
- Luchini C, Bibeau F, Ligtenberg MJL, et al.: ESMO recommendations on microsatellite instability testing for immunotherapy in cancer, and its relationship with PD-1/PD-L1 expression and tumour mutational burden: a systematic review-based approach. Ann Oncol 30 (8): 1232-1243, 2019. [PUBMED Abstract]
- Marcus L, Lemery SJ, Keegan P, et al.: FDA Approval Summary: Pembrolizumab for the Treatment of Microsatellite Instability-High Solid Tumors. Clin Cancer Res 25 (13): 3753-3758, 2019. [PUBMED Abstract]
- Tanizaki J, Yonemori K, Akiyoshi K, et al.: Open-label phase II study of the efficacy of nivolumab for cancer of unknown primary. Ann Oncol 33 (2): 216-226, 2022. [PUBMED Abstract]
- Hellmann MD, Ciuleanu TE, Pluzanski A, et al.: Nivolumab plus Ipilimumab in Lung Cancer with a High Tumor Mutational Burden. N Engl J Med 378 (22): 2093-2104, 2018. [PUBMED Abstract]
- Krämer A, Bochtler T, Pauli C, et al.: Cancer of unknown primary: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol 34 (3): 228-246, 2023. [PUBMED Abstract]
- Huebner G, Link H, Kohne CH, et al.: Paclitaxel and carboplatin vs gemcitabine and vinorelbine in patients with adeno- or undifferentiated carcinoma of unknown primary: a randomised prospective phase II trial. Br J Cancer 100 (1): 44-9, 2009. [PUBMED Abstract]
- Gross-Goupil M, Fourcade A, Blot E, et al.: Cisplatin alone or combined with gemcitabine in carcinomas of unknown primary: results of the randomised GEFCAPI 02 trial. Eur J Cancer 48 (5): 721-7, 2012. [PUBMED Abstract]
- Lee J, Hahn S, Kim DW, et al.: Evaluation of survival benefits by platinums and taxanes for an unfavourable subset of carcinoma of unknown primary: a systematic review and meta-analysis. Br J Cancer 108 (1): 39-48, 2013. [PUBMED Abstract]
- Hainsworth JD, Spigel DR, Clark BL, et al.: Paclitaxel/carboplatin/etoposide versus gemcitabine/irinotecan in the first-line treatment of patients with carcinoma of unknown primary site: a randomized, phase III Sarah Cannon Oncology Research Consortium Trial. Cancer J 16 (1): 70-5, 2010. [PUBMED Abstract]
Follow-Up Treatment of Cancer of Unknown Primary in Remission
The use of follow-up studies including computed tomography or magnetic resonance imaging, is extrapolated from paradigms of other related malignancies with known primary sites. No prospective trials evaluating clinical surveillance have been conducted for patients with cancer of unknown primary.
Current Clinical Trials
Use our advanced clinical trial search to find NCI-supported cancer clinical trials that are now enrolling patients. The search can be narrowed by location of the trial, type of treatment, name of the drug, and other criteria. General information about clinical trials is also available.
Latest Updates to This Summary (05/07/2025)
The PDQ cancer information summaries are reviewed regularly and updated as new information becomes available. This section describes the latest changes made to this summary as of the date above.
Editorial changes were made to this summary.
This summary is written and maintained by the PDQ Adult Treatment Editorial Board, which is editorially independent of NCI. The summary reflects an independent review of the literature and does not represent a policy statement of NCI or NIH. More information about summary policies and the role of the PDQ Editorial Boards in maintaining the PDQ summaries can be found on the About This PDQ Summary and PDQ® Cancer Information for Health Professionals pages.
About This PDQ Summary
Purpose of This Summary
This PDQ cancer information summary for health professionals provides comprehensive, peer-reviewed, evidence-based information about the treatment of cancer of unknown primary. It is intended as a resource to inform and assist clinicians in the care of their patients. It does not provide formal guidelines or recommendations for making health care decisions.
Reviewers and Updates
This summary is reviewed regularly and updated as necessary by the PDQ Adult Treatment Editorial Board, which is editorially independent of the National Cancer Institute (NCI). The summary reflects an independent review of the literature and does not represent a policy statement of NCI or the National Institutes of Health (NIH).
Board members review recently published articles each month to determine whether an article should:
- be discussed at a meeting,
- be cited with text, or
- replace or update an existing article that is already cited.
Changes to the summaries are made through a consensus process in which Board members evaluate the strength of the evidence in the published articles and determine how the article should be included in the summary.
The lead reviewers for Cancer of Unknown Primary (CUP) Treatment are:
- Jacob Adashek, DO (Consultant)
- Eric J. Seifter, MD (Johns Hopkins University)
Any comments or questions about the summary content should be submitted to Cancer.gov through the NCI website’s Email Us. Do not contact the individual Board Members with questions or comments about the summaries. Board members will not respond to individual inquiries.
Levels of Evidence
Some of the reference citations in this summary are accompanied by a level-of-evidence designation. These designations are intended to help readers assess the strength of the evidence supporting the use of specific interventions or approaches. The PDQ Adult Treatment Editorial Board uses a formal evidence ranking system in developing its level-of-evidence designations.
Permission to Use This Summary
PDQ is a registered trademark. Although the content of PDQ documents can be used freely as text, it cannot be identified as an NCI PDQ cancer information summary unless it is presented in its entirety and is regularly updated. However, an author would be permitted to write a sentence such as “NCI’s PDQ cancer information summary about breast cancer prevention states the risks succinctly: [include excerpt from the summary].”
The preferred citation for this PDQ summary is:
PDQ® Adult Treatment Editorial Board. PDQ Cancer of Unknown Primary (CUP) Treatment. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: /types/unknown-primary/hp/unknown-primary-treatment-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389252]
Images in this summary are used with permission of the author(s), artist, and/or publisher for use within the PDQ summaries only. Permission to use images outside the context of PDQ information must be obtained from the owner(s) and cannot be granted by the National Cancer Institute. Information about using the illustrations in this summary, along with many other cancer-related images, is available in Visuals Online, a collection of over 2,000 scientific images.
Disclaimer
Based on the strength of the available evidence, treatment options may be described as either “standard” or “under clinical evaluation.” These classifications should not be used as a basis for insurance reimbursement determinations. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website’s Email Us.