Screening is looking for cancer before a person has any symptoms. This can help detect cancer at an early stage. When abnormal tissue or cancer is found early, it may be easier to treat.
In the United States, there are no standard or routine screening tests to detect stomach cancer in people at average risk. Researchers are working to develop such tests. If a test reduces the number of deaths from stomach cancer, it may become a standard screening test.
Tests to screen for stomach cancer
Some people who have a higher risk of stomach cancer may benefit from screening with upper endoscopy, including:
- older people with chronic gastric atrophy or pernicious anemia
- people who have had
- partial gastrectomy
- a family history of stomach cancer
- people who have certain genetic syndromes
- people from countries where stomach cancer is more common
Learn more about risk factors for stomach cancer.
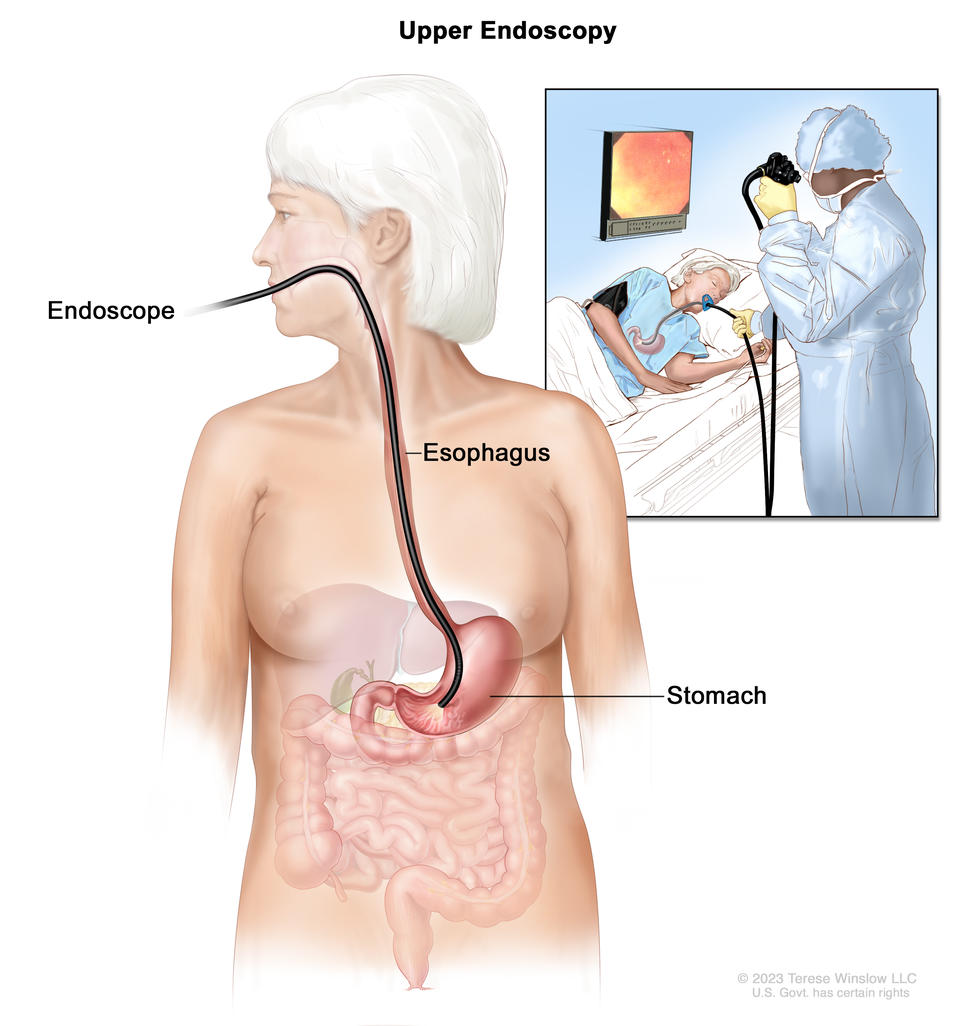
Upper endoscopy
Upper endoscopy is a procedure to look inside the esophagus, stomach, and duodenum (first part of the small intestine) to check for abnormal areas. An endoscope is passed through the mouth and down the throat into the esophagus. An endoscope is a thin, tube-like instrument with a light and a lens for viewing. It may also have a tool to remove tissue, which is checked under a microscope for signs of disease.
Risks of stomach cancer screening
Talk with your doctor about whether stomach cancer screening is right for you.
Potential risks of stomach cancer screening include:
- False-negative test results. Screening test results may appear to be normal even though stomach cancer is present. A person who receives a false-negative test result (one that shows there is no cancer when there really is) may delay seeking medical care even if there are symptoms.
- False-positive test results. Screening test results may appear to be abnormal even though no cancer is present. A false-positive test result (one that shows there is cancer when there really isn’t) can cause anxiety and is usually followed by more tests and procedures which also have risks.
- Risks of the screening test itself. Upper endoscopy may cause the following rare, but serious, complications:
- a small hole (puncture) in the esophagus or stomach
- heart problems
- breathing problems
- lung infection from inhaling food, fluid, or stomach acid into the lung
- severe bleeding that needs to be treated at a hospital
- reactions to medicine used during the procedure