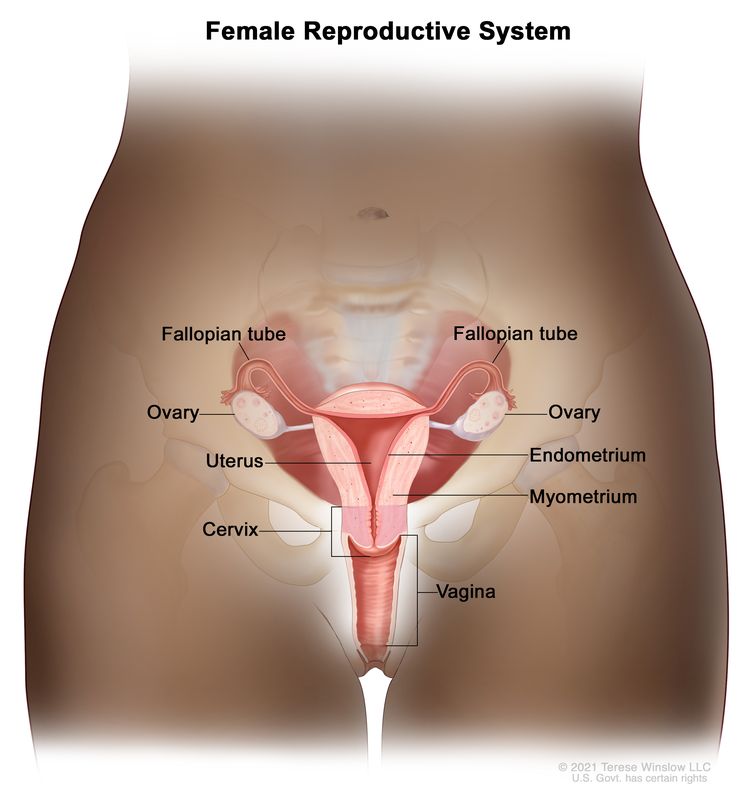
Cervical Cancer Prevention (PDQ®)–Health Professional Version
Overview
Note: The Overview section summarizes the published evidence on this topic. The rest of the summary describes the evidence in more detail.
Other PDQ summaries on Cervical Cancer Screening and Cervical Cancer Treatment are also available.
Who Is at Risk?
Carcinogenic types of human papillomavirus (HPV) are the primary, etiologic, infectious agents that cause virtually all cases of cervical cancer. HPV type 16 (HPV-16) and HPV type 18 (HPV-18) are most often associated with invasive disease.[1,2] Because HPV can be transmitted during sexual activity, there is an association between beginning sexual activity at a younger age, as well as having a greater number of lifetime sexual partners and an increased risk of developing cervical cancer.[3] Immunosuppression is another risk factor for cervical cancer; for example, coinfection with HIV may lead to long-term persistence of viral infection (i.e., failure to clear).[4,5] Once HPV infection occurs, several additional risk factors are associated with a higher risk of the eventual development of cervical cancer. These risk factors include high parity, long-term use of oral contraceptives, and active and passive cigarette smoking.[6–8] The risk increases with longer duration and intensity of smoking. Diethylstilbestrol (DES) exposure in utero is also associated with an increased risk of developing cervical dysplasia.[9]
Factors With Adequate Evidence of an Increased Risk of Cervical Cancer
Human papillomavirus (HPV)
Based on solid evidence from observational studies, HPV infection is associated with the development of cervical cancer.
Magnitude of Effect: HPV has been implicated as the primary etiologic infectious agent causing virtually all cases of cervical cancer.
- Study Design: Evidence obtained from cohort and case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Immunosuppression
Based on solid evidence, being immunosuppressed is associated with an increased risk of cervical cancer.
- Study Design: Evidence obtained from cohort and case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Sexual activity at an early age and with a greater number of partners
Based on solid evidence, sexual activity at a younger age and an increasing number of sexual partners are both associated with an increased risk of HPV infection and subsequent development of cervical cancer.
Magnitude of Effect: Women who experience first sexual intercourse at age 17 years or younger or women who have had six or more lifetime sexual partners have approximately two to three times the risk of squamous cell carcinoma or adenocarcinoma of the cervix, compared with women aged 21 years or older or who have a single sexual partner.[3]
- Study Design: Evidence obtained from cohort and case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
High parity
Based on solid evidence, high parity is associated with increased risk of cervical cancer in HPV-infected women.
Magnitude of Effect: Among HPV-infected women, those who have had seven or more full-term pregnancies have approximately four times the risk of squamous cell cancer compared with nulliparous women, and HPV-infected women also have two to three times the risk of women who have had one or two full-term pregnancies.[6]
- Study Design: Evidence obtained from cohort or case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Long-term use of oral contraceptives
Based on solid evidence, long-term use of oral contraceptives is associated with increased risk of cervical cancer in HPV-infected women.
Magnitude of Effect: Among HPV-infected women, those who used oral contraceptives for 5 to 9 years have approximately three times the incidence of invasive cancer, and those who used them for 10 years or longer have approximately four times the risk.[7]
- Study Design: Evidence obtained from cohort or case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Cigarette smoke exposure
Based on solid evidence, cigarette smoking, both active and passive, is associated with an increased risk of cervical cancer in HPV-infected women.
Magnitude of Effect: Among HPV-infected women, current and former smokers have approximately two to three times the incidence of high-grade cervical intraepithelial neoplasia or invasive cancer. Passive smoking is also associated with increased risk but to a lesser extent.
- Study Design: Evidence obtained from cohort or case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Diethylstilbestrol (DES) exposure
Based on solid evidence, DES exposure is associated with an increased risk of developing clear cell adenocarcinoma of the cervix.
Magnitude of Effect: About one in 1,000 women exposed to DES in utero will develop a clear cell adenocarcinoma of the cervix.
- Study Design: Evidence obtained from cohort studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Factors With Adequate Evidence of a Decreased Risk of Cervical Cancer
Sexual abstinence
Based on solid evidence, abstinence from sexual activity is associated with a near-total reduction in the risk of developing cervical cancer.
Magnitude of Effect: Sexual abstinence essentially precludes HPV transmission.
- Study Design: Evidence obtained from cohort or case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Interventions With Adequate Evidence of a Decreased Risk of Cervical Cancer
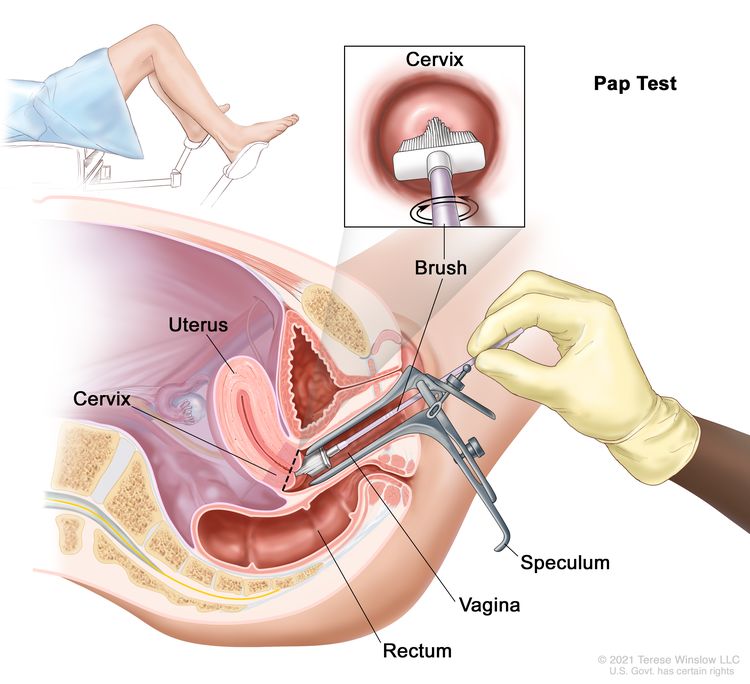
Note: Based on solid evidence, screening with the Papanicolaou (Pap) test and screening with the HPV DNA test reduces cervical cancer incidence. For more information on these screening tests, see Cervical Cancer Screening.
HPV vaccination: Benefits
Based on solid evidence, vaccination against HPV-16/HPV-18 is effective in preventing HPV infection in HPV-naive individuals and is associated with a reduced incidence of cervical intraepithelial neoplasia 2 and 3. By extrapolation, these vaccines should also be associated with a reduced incidence of cervical cancer.
Magnitude of Effect: Vaccination against HPV-16 and HPV-18 reduces incident and persistent infections with efficacy of 91.6% (95% confidence interval [CI], 64.5%–98.0%) and 100% (95% CI, 45%–100%), respectively.
- Study Design: Evidence obtained from randomized controlled trials (for intraepithelial precursor lesions) and cohort study analyses (for invasive cervical cancers).
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
HPV vaccination: Harms
Based on solid evidence, harms of HPV vaccines include injection-site reactions, dizziness and syncope, headache, and fever. Vaccination during pregnancy has not been associated with adverse pregnancy outcomes.[10] Allergic reactions occur rarely.
- Study Design: Evidence obtained from randomized controlled trials.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Use of barrier protection during sexual intercourse: Benefits
Based on solid evidence, the use of barrier methods (e.g., condoms) during sexual intercourse is associated with a decreased risk of cervical cancer.
Magnitude of Effect: Total use of barrier protection decreases cervical cancer incidence (relative risk, 0.4; 95% CI, 0.2–0.9).
- Study Design: Evidence obtained from cohort and case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
Use of barrier protection during sexual intercourse: Harms
Based on fair evidence, the use of barrier methods during sexual intercourse is associated with few serious harms. Barrier methods can break, potentially resulting in unintended pregnancy. Allergic reactions to the barrier material (e.g., natural latex) can occur.
- Study Design: Evidence obtained from cohort and case-control studies.
- Internal Validity: Good.
- Consistency: Good.
- External Validity: Good.
References
- Schiffman M, Castle PE, Jeronimo J, et al.: Human papillomavirus and cervical cancer. Lancet 370 (9590): 890-907, 2007. [PUBMED Abstract]
- Trottier H, Franco EL: The epidemiology of genital human papillomavirus infection. Vaccine 24 (Suppl 1): S1-15, 2006. [PUBMED Abstract]
- Berrington de González A, Green J; International Collaboration of Epidemiological Studies of Cervical Cancer: Comparison of risk factors for invasive squamous cell carcinoma and adenocarcinoma of the cervix: collaborative reanalysis of individual data on 8,097 women with squamous cell carcinoma and 1,374 women with adenocarcinoma from 12 epidemiological studies. Int J Cancer 120 (4): 885-91, 2007. [PUBMED Abstract]
- Abraham AG, D’Souza G, Jing Y, et al.: Invasive cervical cancer risk among HIV-infected women: a North American multicohort collaboration prospective study. J Acquir Immune Defic Syndr 62 (4): 405-13, 2013. [PUBMED Abstract]
- Grulich AE, van Leeuwen MT, Falster MO, et al.: Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. Lancet 370 (9581): 59-67, 2007. [PUBMED Abstract]
- Muñoz N, Franceschi S, Bosetti C, et al.: Role of parity and human papillomavirus in cervical cancer: the IARC multicentric case-control study. Lancet 359 (9312): 1093-101, 2002. [PUBMED Abstract]
- Moreno V, Bosch FX, Muñoz N, et al.: Effect of oral contraceptives on risk of cervical cancer in women with human papillomavirus infection: the IARC multicentric case-control study. Lancet 359 (9312): 1085-92, 2002. [PUBMED Abstract]
- Appleby P, Beral V, Berrington de González A, et al.: Carcinoma of the cervix and tobacco smoking: collaborative reanalysis of individual data on 13,541 women with carcinoma of the cervix and 23,017 women without carcinoma of the cervix from 23 epidemiological studies. Int J Cancer 118 (6): 1481-95, 2006. [PUBMED Abstract]
- Hoover RN, Hyer M, Pfeiffer RM, et al.: Adverse health outcomes in women exposed in utero to diethylstilbestrol. N Engl J Med 365 (14): 1304-14, 2011. [PUBMED Abstract]
- Scheller NM, Pasternak B, Mølgaard-Nielsen D, et al.: Quadrivalent HPV Vaccination and the Risk of Adverse Pregnancy Outcomes. N Engl J Med 376 (13): 1223-1233, 2017. [PUBMED Abstract]
Incidence and Mortality
An estimated 13,360 new cervical cancers and 4,320 cervical cancer deaths will occur in the United States in 2025.[1] When corrected for the prevalence of hysterectomy, the mortality rate for Black women is nearly twice the mortality rate for White women.[2] Also, approximately 1,250,000 women will be diagnosed with precancers annually by cytology using the Papanicolaou (Pap) smear. A continuum of pathological changes may be diagnosed, ranging from atypical squamous cells of undetermined significance to low-grade squamous intraepithelial lesions (LSIL) to high-grade squamous intraepithelial lesions (HSIL) to invasive cancer. The precancerous conditions LSIL and HSIL are also referred to as cervical intraepithelial neoplasia (CIN) 1, 2, and 3. Lesions can regress, persist, or progress to an invasive malignancy, with LSIL (CIN 1) more likely to regress spontaneously and HSIL (CIN 2/CIN 3) more likely to persist or progress. The average time for progression of CIN 3 to invasive cancer has been estimated to be 10 to 15 years.[3]
References
- American Cancer Society: Cancer Facts and Figures 2025. American Cancer Society, 2025. Available online. Last accessed January 16, 2025.
- Beavis AL, Gravitt PE, Rositch AF: Hysterectomy-corrected cervical cancer mortality rates reveal a larger racial disparity in the United States. Cancer 123 (6): 1044-1050, 2017. [PUBMED Abstract]
- Holowaty P, Miller AB, Rohan T, et al.: Natural history of dysplasia of the uterine cervix. J Natl Cancer Inst 91 (3): 252-8, 1999. [PUBMED Abstract]
Factors With Adequate Evidence of an Increased Risk of Cervical Cancer
HPV
Epidemiological studies to evaluate risk factors for the development of squamous intraepithelial lesions and cervical malignancy demonstrate conclusively a sexual mode of transmission of a carcinogen.[1] It is now widely accepted that human papillomavirus (HPV) is the primary etiologic infectious agent that causes virtually all cases of cervical cancer.[2,3] Other sexually transmitted factors, including herpes simplex virus 2 and Chlamydia trachomatis, may play a co-causative role.[4] More than 80 distinct types of HPV have been identified, approximately 30 of which infect the human genital tract. HPV type 16 (HPV-16) and HPV type 18 (HPV-18) are most often associated with invasive disease. Characterization of carcinogenic risk associated with HPV types is an important step in the process of developing a combination HPV vaccine for the prevention of cervical neoplasia. In a population-based study of HPV infection and cervical neoplasia in Costa Rica, 80% of high-grade squamous intraepithelial lesions (HSIL) and invasive lesions were associated with HPV infection by one or more of 13 cancer-associated types.[5] In this study, the risk of about 50% of HSIL and invasive cervical cancer was attributable to HPV-16. HPV-18 was associated with 15% of invasive disease but only 5% of HSIL, suggesting that HPV-18 may have a role in more aggressive cases of cervical malignancy. There may be differences in the prevalence and carcinogenic risk of individual high-risk HPV genotypes by race and geographical regions, and additional studies are ongoing.[6]
Immunosuppression
Most cases of HPV infection are resolved by the host immune system. Immunosuppression leads to persistence of viral infection with a subsequent increased risk of cervical neoplasia. Women with immunosuppression resulting from HIV infection have been studied over the past three decades of the AIDS epidemic. In one North American study, a group of 13,690 HIV-infected women were studied for a median of 5 years. The rate of invasive cervical cancer in the HIV-infected women was 26 cases per 100,000 women, and this was approximately four times greater than an HIV-uninfected control group.[7] HIV-infected women with the lowest CD4 lymphocyte counts were at the highest risk of invasive cancer. Women who are immunosuppressed resulting from organ transplantation are also at risk of invasive cervical cancer, and one meta-analysis found a twofold increased risk.[8]
Sexual Activity at an Early Age and With a Greater Number of Partners
HPV infection has been established as a necessary cause of almost all cases of cervical cancer, and the primary mode of transmission is sexual contact. This provides context for the findings that younger age at first intercourse and an increasing number of lifetime sexual partners are both associated with an increased risk of developing cervical cancer. Pooled, individual, patient-level data from 12 cohort and case-control studies demonstrated statistically significantly increased risks of developing cervical cancer in women who were aged 17 years or younger at first intercourse, compared with women who were aged 21 years or older at first intercourse (relative risk [RR] for squamous cell cancer, 2.24; 95% confidence interval [CI], 2.11–2.38 and RR for adenocarcinoma, 2.06; 95% CI, 1.83–2.33). Similar findings were observed in women who had six or more lifetime sexual partners, compared with women who had one lifetime sexual partner (RR for squamous cell cancer, 2.98; 95% CI, 2.62–3.40 and RR for adenocarcinoma, 2.64; 95% CI, 2.07–3.36).[9]
High Parity
High parity has long been recognized as a risk factor for cervical cancer, but the relation of parity to HPV infection was uncertain. A meta-analysis of 25 epidemiological studies, including 16,563 women with cervical cancer and 33,542 women without cervical cancer, showed that the number of full-term pregnancies was associated with increased risk, regardless of age at first pregnancy. This finding was also true if analyses were limited to patients with high-risk HPV infections (RR, 4.99; 95% CI, 3.49–7.13 for seven or more pregnancies vs. no pregnancies; linear trend test X2 = 30.69; P < .001).[10]
Long-Term Use of Oral Contraceptives
Long-term use of oral contraceptives has also been known to be associated with cervical cancer, but its relation to HPV infection was also uncertain. A pooled analysis of HPV-positive women from the studies described above was undertaken. Compared with women who have never used oral contraceptives, those who have used them for fewer than 5 years did not have an increased risk of cervical cancer (odds ratio [OR], 0.73; 95% CI, 0.52–1.03). The OR for women who used oral contraceptives for 5 to 9 years was 2.82 (95% CI, 1.46–5.42), and for 10 or more years, the OR was 4.03 (95% CI, 2.09–8.02).[11] A meta-analysis of 24 epidemiological studies confirmed the increased risk associated with oral contraceptives, which is proportionate to the duration of use. Risk decreases after cessation and returns to normal risk levels in 10 years.[12]
Cigarette Smoke Exposure
Cigarette smoking by women is associated with an increased risk of squamous cell carcinoma.[1,13,14] This risk increases with longer duration and intensity of smoking. The risk among smokers may be present with exposure to environmental tobacco smoke and may be as high as four times that of women who are nonsmokers who are not exposed to environmental smoking.[1] Case-control studies of women infected with HPV have examined the effect of various types and levels of tobacco exposure and found similar results.[14–16]
DES Exposure
Diethylstilbestrol (DES) is a synthetic form of estrogen that was prescribed to pregnant women in the United States between 1940 and 1971 to prevent miscarriage and premature labor. DES is associated with a substantially increased risk of developing clear cell adenocarcinoma of the vagina and cervix among the daughters of women who used the drug during pregnancy (standardized incidence ratio, 24.23; 95% CI, 8.89–52.74); the risk persists as these women age into their 40s.[17] Despite the greatly elevated risk relative to the general population, this type of cancer is still rare; about one in 1,000 daughters exposed to DES will develop a clear cell adenocarcinoma.
DES exposure in utero is also associated with an increased risk of developing cervical dysplasia. An evaluation of three cohorts, including the Diethylstilbestrol Adenosis study, the Dieckmann study, and the Women’s Health Study, with long-term follow-up of more than 4,500 women exposed in utero to DES, found that 6.9% of exposed women developed grade II or higher cervical intraepithelial neoplasia, compared with 3.4% of nonexposed women (hazard ratio, 2.28; 95% CI, 1.59–3.27).[18]
References
- Brinton LA: Epidemiology of cervical cancer–overview. IARC Sci Publ (119): 3-23, 1992. [PUBMED Abstract]
- Schiffman M, Castle PE, Jeronimo J, et al.: Human papillomavirus and cervical cancer. Lancet 370 (9590): 890-907, 2007. [PUBMED Abstract]
- Trottier H, Franco EL: The epidemiology of genital human papillomavirus infection. Vaccine 24 (Suppl 1): S1-15, 2006. [PUBMED Abstract]
- Ault KA: Epidemiology and natural history of human papillomavirus infections in the female genital tract. Infect Dis Obstet Gynecol 2006 (Suppl): 40470, 2006. [PUBMED Abstract]
- Herrero R, Hildesheim A, Bratti C, et al.: Population-based study of human papillomavirus infection and cervical neoplasia in rural Costa Rica. J Natl Cancer Inst 92 (6): 464-74, 2000. [PUBMED Abstract]
- Risley C, Clarke MA, Geisinger KR, et al.: Racial differences in HPV type 16 prevalence in women with ASCUS of the uterine cervix. Cancer Cytopathol 128 (8): 528-534, 2020. [PUBMED Abstract]
- Abraham AG, D’Souza G, Jing Y, et al.: Invasive cervical cancer risk among HIV-infected women: a North American multicohort collaboration prospective study. J Acquir Immune Defic Syndr 62 (4): 405-13, 2013. [PUBMED Abstract]
- Grulich AE, van Leeuwen MT, Falster MO, et al.: Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. Lancet 370 (9581): 59-67, 2007. [PUBMED Abstract]
- Berrington de González A, Green J; International Collaboration of Epidemiological Studies of Cervical Cancer: Comparison of risk factors for invasive squamous cell carcinoma and adenocarcinoma of the cervix: collaborative reanalysis of individual data on 8,097 women with squamous cell carcinoma and 1,374 women with adenocarcinoma from 12 epidemiological studies. Int J Cancer 120 (4): 885-91, 2007. [PUBMED Abstract]
- International Collaboration of Epidemiological Studies of Cervical Cancer: Cervical carcinoma and reproductive factors: collaborative reanalysis of individual data on 16,563 women with cervical carcinoma and 33,542 women without cervical carcinoma from 25 epidemiological studies. Int J Cancer 119 (5): 1108-24, 2006. [PUBMED Abstract]
- Moreno V, Bosch FX, Muñoz N, et al.: Effect of oral contraceptives on risk of cervical cancer in women with human papillomavirus infection: the IARC multicentric case-control study. Lancet 359 (9312): 1085-92, 2002. [PUBMED Abstract]
- Appleby P, Beral V, Berrington de González A, et al.: Cervical cancer and hormonal contraceptives: collaborative reanalysis of individual data for 16,573 women with cervical cancer and 35,509 women without cervical cancer from 24 epidemiological studies. Lancet 370 (9599): 1609-21, 2007. [PUBMED Abstract]
- Hellberg D, Nilsson S, Haley NJ, et al.: Smoking and cervical intraepithelial neoplasia: nicotine and cotinine in serum and cervical mucus in smokers and nonsmokers. Am J Obstet Gynecol 158 (4): 910-3, 1988. [PUBMED Abstract]
- Brock KE, MacLennan R, Brinton LA, et al.: Smoking and infectious agents and risk of in situ cervical cancer in Sydney, Australia. Cancer Res 49 (17): 4925-8, 1989. [PUBMED Abstract]
- Ho GY, Kadish AS, Burk RD, et al.: HPV 16 and cigarette smoking as risk factors for high-grade cervical intra-epithelial neoplasia. Int J Cancer 78 (3): 281-5, 1998. [PUBMED Abstract]
- Plummer M, Herrero R, Franceschi S, et al.: Smoking and cervical cancer: pooled analysis of the IARC multi-centric case–control study. Cancer Causes Control 14 (9): 805-14, 2003. [PUBMED Abstract]
- Verloop J, van Leeuwen FE, Helmerhorst TJ, et al.: Cancer risk in DES daughters. Cancer Causes Control 21 (7): 999-1007, 2010. [PUBMED Abstract]
- Hoover RN, Hyer M, Pfeiffer RM, et al.: Adverse health outcomes in women exposed in utero to diethylstilbestrol. N Engl J Med 365 (14): 1304-14, 2011. [PUBMED Abstract]
Factors With Adequate Evidence of a Decreased Risk of Cervical Cancer
Sexual Abstinence
Nearly all cases of cervical cancer are associated with human papillomavirus (HPV) infection, which is transmitted during sexual activity. Therefore, cervical cancer is seen more frequently in women with sexual activity at an early age and with multiple partners.[1] Lifetime abstinence from sexual activity is associated with a near-total reduction in the risk of developing cervical cancer. For more information, see the HPV section.
References
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans: Human papillomaviruses. IARC Monogr Eval Carcinog Risks Hum 100 (Pt B), 255-296, 2012. Available online. Last accessed January 31, 2025.
Interventions With Adequate Evidence of a Decreased Risk of Cervical Cancer
HPV Vaccination
Given the etiological role of human papillomavirus (HPV) in the pathogenesis of cervical neoplasia, vaccines to immunize individuals against HPV infection offer a primary prevention strategy for cervical cancer. A quadrivalent (HPV type 6 [HPV-6], type 11 [HPV-11], type 16 [HPV-16], and type 18 [HPV-18]) vaccine using a late protein L1 construct to induce antibody-mediated immunity was approved for use by the U.S. Food and Drug Administration in 2006; a bivalent (HPV-16, -18) vaccine was approved in 2009; and a vaccine targeting nine HPV types was approved in 2014. Vaccination during pregnancy has not been associated with adverse pregnancy outcomes.[1]
Persistent infection with oncogenic types of HPV, such as HPV-16 and HPV-18, is associated with the development of cervical cancer.[2] A vaccine to prevent HPV infection with oncogenic-type viruses has the potential to reduce the incidence of cervical cancer. A vaccine against HPV-16 using empty-viral capsids called virus-like particles (VLP) was developed and tested for efficacy in preventing persistent infection with HPV-16.
A multicenter, double-blind, placebo-controlled trial enrolled 2,391 women aged 16 to 23 years and randomly assigned them to receive either 40 µg of HPV-16 L1 VLP vaccine or placebo on day 1, at 2 months, and at 6 months. Papanicolaou (Pap) tests and genital samples for HPV-16 DNA were obtained on day 1, at 7 months, and every 6 months for 48 months. Colposcopy and cervical biopsies were obtained when clinically indicated at study exit. Serum HPV-16 antibody titers were obtained at study entry, at 7 months, and then every 6 months. A total of 1,505 women (755 receiving vaccine and 750 receiving placebo) completed all three vaccinations and had follow-up after month 7. After immunization, HPV titers peaked at month 7, declined through month 18, and then stabilized in months 30 through 48. There were no cases of cervical intraepithelial neoplasia (CIN) in the vaccine-treated women, but there were 12 cases in the placebo group (six CIN 2 and six CIN 3). HPV-16 infection that persisted for at least 4 months was seen in seven vaccine-treated women, compared with 111 placebo-treated women.[3]
An international, double-blind, placebo-controlled trial of a bivalent HPV-16/HPV-18 VLP vaccine was performed in 1,113 women aged 15 to 25 years with normal cervical cytology who were seronegative for HPV-16, HPV-18, and 12 other oncogenic HPV types at enrollment. Women received either vaccine or placebo at 0, 1, and 6 months and were assessed by cervical cytology and self-obtained cervicovaginal samples for at least 18 months. A masked treatment-allocation follow-up study was performed for an additional 3 years, for a combined analysis of up to 6.4 years of follow-up. The 12-month persistent infection rate of HPV-16 or HPV-18 in an according-to-protocol cohort (i.e., women who received all three doses of vaccine or placebo on the correct schedule) was 0 of 401 women in the vaccine arm, compared with 20 of 372 women in the placebo arm, with a vaccine efficacy rate of 100% (95% confidence interval [CI], 81.8%–100%). Diagnoses of CIN 2 or higher in a total vaccinated cohort (i.e., women who received at least one dose of vaccine or placebo) were 0 of 481 women in the vaccine arm compared with 9 of 470 women in the placebo arm, with a vaccine efficacy of 100% (95% CI, 51.3%–100%). Adverse events were similar in vaccinated and placebo-treated women. Neither analysis was intention-to-treat (ITT), making it difficult to know what the true vaccine efficacy for either virological or cytohistological end points would be in the routine clinical setting. Furthermore, cytohistological outcomes were reported only as composite end points (CIN 2+), making it impossible to distinguish the vaccine’s efficacy against invasive cervical cancer alone and potentially inflating the observed efficacy by including lesions with a relatively high probability (approximately 50% for CIN 2 [4]) of spontaneous regression.[5] A register-based observational study in England reported the impact of a national bivalent vaccination program on cervical cancer and CIN 3.[6] Routine vaccinations were offered to girls aged 12 to 13 years with a catch-up program for those aged 14 to 18 years. Data from 13.7 million years of follow-up in women aged 20 years to younger than 30 years showed a substantial reduction in cervical cancer and CIN 3 incidence after a national HPV program was introduced. This was especially true in individuals who were offered the vaccine between ages 12 and 13 years (see Table 1).
| Estimated Relative Reduction (95% CI) | ||
|---|---|---|
| Age Vaccine Was Offered | Cervical Cancer | CIN 3 |
| CI = confidence interval; CIN = cervical intraepithelial neoplasia. | ||
| 16–18 y | 34% (25–41) | 39% (36–41) |
| 14–16 y | 62% (52–71) | 75% (72–77) |
| 12–13 y | 87% (72–94) | 97% (96–78) |
A quadrivalent vaccine (HPV types-6, -11, -16, and -18) was evaluated in a multinational, double-blind, randomized controlled trial of 17,622 women aged 15 to 26 years (FUTURE I and II).[7] Women received either the HPV vaccine or placebo at 0, 2, and 6 months; participants were assessed by clinical exam, Pap test, and HPV DNA testing for 4 or more years. Two analyses were reported. One group was considered to be HPV naive: negative to 14 HPV types. The second group was an ITT analysis, which approximates a sexually active population. The composite end point for cervical disease included the incidence of HPV-16/HPV-18–related, CIN 2, CIN 3, adenocarcinoma in situ, or invasive carcinoma. Outcomes were reported as follows:
| Population | Point Estimate and 95% CI |
|---|---|
| CI = confidence interval; CIN = cervical intraepithelial neoplasia; HPV = human papillomavirus; ITT = intention-to-treat. | |
| HPV-naive population for HPV-CIN 3 | 100% (90.5%–100%) for lesions associated with HPV-6, -11, -16, or -18 |
| ITT CIN 3 | 45.3% (29.8%–57.6%) for lesions associated with HPV-6, -11, -16, or -18 |
This study also demonstrated decreased rates of abnormal Pap tests and subsequent diagnostic procedures. No cases of invasive cervical cancer were identified during the trial.
A 9-valent HPV (9vHPV) vaccine was studied in another international randomized trial, which included 14,215 women. This new vaccine, 9vHPV, includes the four HPV types in the quadrivalent vaccine, qHPV (6, 11, 16, 18) and also 5 more oncogenic types (31, 33, 45, 52, 58). Sexually active women aged 16 to 26 years with fewer than five lifetime sexual partners received three intramuscular injections (day 1, month 2, and month 6) of either the qHPV vaccine or the 9vHPV vaccine. Women were evaluated every 6 months for up to 5 years. The rate of high-grade cervical, vulvar, or vaginal disease was the same in both groups (14.0 per 1,000 person-years) because of preexisting HPV infection, but the rate of disease related to HPV-31, -35, -45, -52, and -58 was lower in the 9vHPV vaccine group (0.1 vs. 1.6 per 1,000 person-years). Injection-site reactions were more common in the 9vHPV group.[8] Although not addressed in this study, the benefit of HPV vaccination is optimal in younger females before the onset of sexual activity.
All forms of the HPV vaccine are currently recommended by the Centers for Disease Control and Prevention (CDC) in the United States as a two-dose schedule at least 6 months apart for adolescents younger than 15 years. The current CDC recommendation for older individuals is to receive the original three-dose series. Recently, given issues of cost and adherence, there has been published data investigating whether similar vaccine efficacy could be obtainable using a reduced-dose schedule. A post hoc combined analysis of two phase III randomized controlled trials of the bivalent HPV vaccine (the Costa Rica Vaccine Trial and the PApilloma TRIal against Cancer In young Adults [PATRICIA] Trial) found that among women who were not HPV positive at enrollment for the specific virus type being studied, vaccine efficacy against either one-time incident detection of HPV 16/18 or incident infection that persisted at least 6 months was not statistically significantly different for those who received all three, two, or only one of the scheduled HPV vaccine doses (resulting from nonadherence or other factors) for up to 4 years of follow-up. Vaccine efficacy rates for persistent HPV 16/18 infection ranged from 89.1% (95% CI, 86.8%–91.0%) for three doses, to 89.7% (95% CI, 73.3%–99.8%) for two doses, to 96.6% (95% CI, 81.7%–99.8%) for one dose. To date, there are no randomized controlled trials that directly assess this clinical question.[9] A recent international study compared a two-dose schedule with a three-dose schedule in adolescents younger than 15 years who received the 9vHPV vaccine. The antibody response was noninferior in the two-dose schedule, leading to the current recommendation that two doses are sufficient for this age group.[10] Long-term follow-up data from the Costa Rica Vaccine Trial suggested that all HPV-vaccinated women aged 18 to 25 years at the time of initial vaccination remained HPV-16/HPV-18 seropositive more than a decade after initial vaccination, regardless of the number of doses received. The antibody levels were lower in the women who received only one dose than in the women who received two or three doses of the bivalent vaccine, but the levels remained higher than estimated levels achieved via natural immunity. The long-term vaccine efficacy rates against prevalent HPV-16 or HPV-18 infection were 80.2% (95% CI, 70.7%–87.0%) among women who received three doses, 83.8% (95% CI, 19.5%–99.2%) among those who received two doses, and 82.1% (95% CI, 40.2%–97.0%) among those who received one dose.[11] Additionally, there was prolonged efficacy against CIN 2 and CIN 3 after 7 to 11 years of follow-up.[12] The women in this long-term follow-up study were not randomly assigned to one, two, or three doses, and the number of women who received only one dose is low. However, the promising findings of the long-term stability of HPV-antibody levels and vaccine efficacy in women who were older than the recommended age at the time of initial vaccination has influenced the design of a currently ongoing trial, which will answer the question of the efficacy of a single dose more definitively. The ESCUDDO study (NCT03180034) is a trial enrolling adolescent girls who will be randomly assigned to either one dose or two doses of the bivalent or nonavalent vaccines. A concurrent population survey of comparable, unvaccinated, age-matched females in the same region will be used for comparison. Results are anticipated in 2025.
On the basis of their mechanism of action, L1/2 HPV vaccines do not appear to impact preexisting infections. The FUTURE II trial demonstrated a markedly lower vaccine efficacy rate in the total randomized study population, which included individuals who were positive for HPV at baseline, compared with the per-protocol population (44% for lesions associated with HPV-16 or HPV-18, and 17% for lesions associated with any HPV type vs. 98%; see Table 2 above).[7] Additionally, an intermediate analysis of a randomized controlled trial primarily evaluating the efficacy of the HPV-16/18 vaccine in preventing infection found no effect on viral clearance rates in women aged 18 to 25 years who were positive at the time of study enrollment.[13]
The type-specific vaccines, if successful in preventing invasive cancer, will offer protection for only a subset of cases, the proportion of which will vary worldwide.[14] Using data from a multicenter case-control study conducted in 25 countries, it was estimated that a vaccine containing the seven most common HPV types could prevent 87% of cervical cancers worldwide. A vaccine with the two most common strains, HPV-16 and HPV-18, would prevent 71% of cervical cancers worldwide.[14]
There is growing evidence of population-level impacts and herd immunity with HPV vaccination. There are data that explore the impact of national HPV vaccination programs and report on vaccine effectiveness. These data come from studies conducted in different countries throughout the world including England, Denmark, Australia, Costa Rica, and the United States. In England, 15,459 residual genital specimens from women aged 16 to 24 years, collected for Chlamydia trachomatis screening between 2010 and 2016, were utilized for national HPV surveillance.[15] In this study, vaccine effectiveness for HPV-16/HPV-18 was 82% (95% CI, 60.6%–91.8%) for women who were vaccinated before age 15 years. Within the younger age groups, the prevalence of HPV-16/HPV-18 significantly decreased within the postvaccination period between 2010 and 2011 to 2016 from 8.2% to 1.6% in 16 to 18 year olds and from 14.0% to 1.6% in 19 to 21 year olds (compared with 17.6% and 16.9% in the prevaccination era).[15] Data from a Danish nationwide cohort study reported the dose-related effectiveness of the quadrivalent HPV vaccine.[16] In this cohort of 590,083 women aged 17 to 25 years, 215,309 (36%) women were vaccinated at age 16 years or younger, and 40,742 (19%) women received less than three doses. Cervical cancer screening rates were similar in the vaccinated and unvaccinated cohorts. In the overall cohort, there were 5,561 cases of CIN 3+ during the follow-up period. Only 5% of cases were in vaccinated women. The incidence rate was 355 cases per 100,000 person-years in unvaccinated women compared with 41 cases per 100,000 person-years in vaccinated women. The incidence rate was independent of the number of vaccine doses administered (incidence rates 40, 41, 40 cases per 100,000 person-years for 1, 2 and 3 doses, respectively).[16]
A study of cervical HPV DNA among 202 Australian women aged 18 to 24 years who were sampled between 2005 and 2007, before implementation of a national quadrivalent prophylactic HPV vaccine program, compared the results with a matched group of 1,058 women who were sampled in the postvaccination era (2010–2012). This study found an adjusted prevalence ratio (PR) among fully vaccinated women of 0.07 (95% CI, 0.04–0.14; P < .0001) for vaccine-related HPV types and a smaller but statistically significant magnitude of protection of 0.65 (95% CI, 0.43–0.96; P < .03) among unvaccinated women, suggesting herd immunity (protection of unvaccinated individuals).[17] These data strengthen previous results that suggest herd immunity in this population, manifested as a reduction in genital warts among heterosexual men, a group that includes sexual partners of vaccinated women.[18] Data also suggest cross protection against carcinogenic types that are not directly targeted by the quadrivalent vaccine but are included in the new nonvalent HPV vaccine.[17] Pooled data from the Costa Rica Vaccine Trial and PATRICIA Study showed that the AS04-adjuvanted HPV-16/HPV-18 vaccine provides additional cross protection beyond established protected types (e.g., 34/35/39/40/42/43/44/51/52/53/54/56/58/59/66/68/73/70/74; efficacy 9/9%; 95% CI, 1.7%–1.4%). This may partially explain the high efficacy of the AS04-adjuvanted HPV-16/HPV-18 vaccine against CIN 3+ (87.8%; 95% CI, 71.1%–95.7%).[19] A meta-analysis that included data from 14 high-income countries with cumulated data from more than 60 million individuals over 8 years reported an 83% decrease in prevalence of HPV-16 and HPV-18 (RR, 0.17; 95% CI, 0.11–0.25) among girls aged 13 to 19 years. There was also evidence of benefit to a more proximal cancer end point. After 5 to 9 years of HPV vaccination, decreased risk of CIN 2+ was also identified among screened girls aged 15 to 19 years (RR, 0.49; 95% CI, 0.42–0.58), and among women aged 20 to 24 years (RR, 0.69; 95% CI, 0.57–0.84) in comparison to an increase seen among screened and mostly unvaccinated women aged 25 to 29 years (RR, 1.19; 95% CI, 1.06–1.32) and aged 30 to 39 years (RR, 1.23; 95% CI, 1.13–1.34).[20]
Data from the National Health and Nutrition Examination Survey (NHANES) from 2003 to 2018 demonstrated an increasing impact of the HPV vaccination program and herd protection in the United States.[21] Overall, the data demonstrated an increase in HPV vaccination coverage among sexually experienced females and males. Importantly, vaccination before age 15 years also increased from 2011 to 2014 and from 2015 to 2018 in both females (27.2%, 48.6%, respectively) and males (18.6%, 48.7%, respectively). From 2015 to 2018, the 4-valent HPV (4vHPV)-type prevalence among sexually experience females aged 14 to 24 years was 85% overall, 90% in vaccinated females, and 74% in unvaccinated females. Estimates of the vaccine’s effectiveness and its impact among vaccinated females were similar from 2007 to 2010 (64% and 61%, respectively) and from 2011 to 2014 (84% and 89%, respectively). However, in 2015 to 2018, these statistics diverged (60% and 90%, respectively). This indicates that as herd protection increases and prevalence among unvaccinated individuals decreases, vaccine effectiveness can be difficult to estimate (1-prevalence ratio between vaccinated and unvaccinated individuals x 100). In 2013 to 2016, the prevalence of 4vHPV types was 1.8% in sexually experienced males who were vaccinated and 3.5% in sexually experienced males who were unvaccinated (PR, 0.49; 95% CI, 0.11–2.20), resulting in an estimated vaccine effectiveness of 51%. Significant declines were not observed in non–4vHPV-type prevalence for females or males. Although notable limitations of this survey study included self-report of HPV vaccine and dose, small sample sizes, and estimates of the impact and effectiveness based on history of at least one vaccine dose, this nationally representative data reflects an increasing impact of the U.S. vaccination program and herd protection.
Association of HPV vaccination with reduced incidence of invasive cervical cancer
In a nationwide population-based cohort study of the impact of the national vaccination program in Sweden using quadrivalent vaccine, more than 1.67 million women aged 10 to 30 years with no previous history of HPV vaccination were followed through the national registry using individual person linkage.[22] The cumulative risk of cervical cancer by age 30 years was 47 cases per 100,000 in vaccinated women, compared with 94 cases per 100,000 in unvaccinated women (incidence rate ratio [IRR], 0.51; 95% CI, 0.32–0.82, adjusting only for age at follow-up). After adjusting for all collected potential confounding factors, the IRR for women vaccinated before age 17 years was 0.12 (95% CI, 0.00–0.34).
Use of Barrier Protection During Sexual Intercourse
Barrier methods of contraception are associated with a reduced incidence of squamous intraepithelial lesions (SIL) presumptively secondary to protection from sexually transmitted disease.[23,24] The effectiveness of condom use for the prevention of HPV infections has been evaluated in a prospective study of women aged 18 to 22 years who were virgins.[25] The number of vulvovaginal HPV infections was reduced with consistent condom use, and the HPV infection rate was 37.8 infections per 100 patient-years among women whose partners used condoms 100% of the time in the 8 months before testing, compared with 89.3 infections per 100 patient-years among women whose partners used condoms less than 5% of the time (P trend = .005). No cervical SIL were detected among women reporting 100% condom use by their partner.[25]
References
- Scheller NM, Pasternak B, Mølgaard-Nielsen D, et al.: Quadrivalent HPV Vaccination and the Risk of Adverse Pregnancy Outcomes. N Engl J Med 376 (13): 1223-1233, 2017. [PUBMED Abstract]
- Wallin KL, Wiklund F, Angström T, et al.: Type-specific persistence of human papillomavirus DNA before the development of invasive cervical cancer. N Engl J Med 341 (22): 1633-8, 1999. [PUBMED Abstract]
- Mao C, Koutsky LA, Ault KA, et al.: Efficacy of human papillomavirus-16 vaccine to prevent cervical intraepithelial neoplasia: a randomized controlled trial. Obstet Gynecol 107 (1): 18-27, 2006. [PUBMED Abstract]
- Castle PE, Schiffman M, Wheeler CM, et al.: Evidence for frequent regression of cervical intraepithelial neoplasia-grade 2. Obstet Gynecol 113 (1): 18-25, 2009. [PUBMED Abstract]
- Romanowski B, de Borba PC, Naud PS, et al.: Sustained efficacy and immunogenicity of the human papillomavirus (HPV)-16/18 AS04-adjuvanted vaccine: analysis of a randomised placebo-controlled trial up to 6.4 years. Lancet 374 (9706): 1975-85, 2009. [PUBMED Abstract]
- Falcaro M, Castañon A, Ndlela B, et al.: The effects of the national HPV vaccination programme in England, UK, on cervical cancer and grade 3 cervical intraepithelial neoplasia incidence: a register-based observational study. Lancet 398 (10316): 2084-2092, 2021. [PUBMED Abstract]
- FUTURE II Study Group: Quadrivalent vaccine against human papillomavirus to prevent high-grade cervical lesions. N Engl J Med 356 (19): 1915-27, 2007. [PUBMED Abstract]
- Joura EA, Giuliano AR, Iversen OE, et al.: A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N Engl J Med 372 (8): 711-23, 2015. [PUBMED Abstract]
- Kreimer AR, Struyf F, Del Rosario-Raymundo MR, et al.: Efficacy of fewer than three doses of an HPV-16/18 AS04-adjuvanted vaccine: combined analysis of data from the Costa Rica Vaccine and PATRICIA trials. Lancet Oncol 16 (7): 775-86, 2015. [PUBMED Abstract]
- Iversen OE, Miranda MJ, Ulied A, et al.: Immunogenicity of the 9-Valent HPV Vaccine Using 2-Dose Regimens in Girls and Boys vs a 3-Dose Regimen in Women. JAMA 316 (22): 2411-2421, 2016. [PUBMED Abstract]
- Kreimer AR, Sampson JN, Porras C, et al.: Evaluation of Durability of a Single Dose of the Bivalent HPV Vaccine: The CVT Trial. J Natl Cancer Inst 112 (10): 1038-1046, 2020. [PUBMED Abstract]
- Porras C, Tsang SH, Herrero R, et al.: Efficacy of the bivalent HPV vaccine against HPV 16/18-associated precancer: long-term follow-up results from the Costa Rica Vaccine Trial. Lancet Oncol 21 (12): 1643-1652, 2020. [PUBMED Abstract]
- Hildesheim A, Herrero R, Wacholder S, et al.: Effect of human papillomavirus 16/18 L1 viruslike particle vaccine among young women with preexisting infection: a randomized trial. JAMA 298 (7): 743-53, 2007. [PUBMED Abstract]
- Muñoz N, Bosch FX, Castellsagué X, et al.: Against which human papillomavirus types shall we vaccinate and screen? The international perspective. Int J Cancer 111 (2): 278-85, 2004. [PUBMED Abstract]
- Mesher D, Panwar K, Thomas SL, et al.: The Impact of the National HPV Vaccination Program in England Using the Bivalent HPV Vaccine: Surveillance of Type-Specific HPV in Young Females, 2010-2016. J Infect Dis 218 (6): 911-921, 2018. [PUBMED Abstract]
- Verdoodt F, Dehlendorff C, Kjaer SK: Dose-related Effectiveness of Quadrivalent Human Papillomavirus Vaccine Against Cervical Intraepithelial Neoplasia: A Danish Nationwide Cohort Study. Clin Infect Dis 70 (4): 608-614, 2020. [PUBMED Abstract]
- Tabrizi SN, Brotherton JM, Kaldor JM, et al.: Assessment of herd immunity and cross-protection after a human papillomavirus vaccination programme in Australia: a repeat cross-sectional study. Lancet Infect Dis 14 (10): 958-66, 2014. [PUBMED Abstract]
- Donovan B, Franklin N, Guy R, et al.: Quadrivalent human papillomavirus vaccination and trends in genital warts in Australia: analysis of national sentinel surveillance data. Lancet Infect Dis 11 (1): 39-44, 2011. [PUBMED Abstract]
- Tota JE, Struyf F, Hildesheim A, et al.: Efficacy of AS04-Adjuvanted Vaccine Against Human Papillomavirus (HPV) Types 16 and 18 in Clearing Incident HPV Infections: Pooled Analysis of Data From the Costa Rica Vaccine Trial and the PATRICIA Study. J Infect Dis 223 (9): 1576-1581, 2021. [PUBMED Abstract]
- Drolet M, Bénard É, Pérez N, et al.: Population-level impact and herd effects following the introduction of human papillomavirus vaccination programmes: updated systematic review and meta-analysis. Lancet 394 (10197): 497-509, 2019. [PUBMED Abstract]
- Rosenblum HG, Lewis RM, Gargano JW, et al.: Human Papillomavirus Vaccine Impact and Effectiveness Through 12 Years After Vaccine Introduction in the United States, 2003 to 2018. Ann Intern Med 175 (7): 918-926, 2022. [PUBMED Abstract]
- Lei J, Ploner A, Elfström KM, et al.: HPV Vaccination and the Risk of Invasive Cervical Cancer. N Engl J Med 383 (14): 1340-1348, 2020. [PUBMED Abstract]
- Parazzini F, Negri E, La Vecchia C, et al.: Barrier methods of contraception and the risk of cervical neoplasia. Contraception 40 (5): 519-30, 1989. [PUBMED Abstract]
- Hildesheim A, Brinton LA, Mallin K, et al.: Barrier and spermicidal contraceptive methods and risk of invasive cervical cancer. Epidemiology 1 (4): 266-72, 1990. [PUBMED Abstract]
- Winer RL, Hughes JP, Feng Q, et al.: Condom use and the risk of genital human papillomavirus infection in young women. N Engl J Med 354 (25): 2645-54, 2006. [PUBMED Abstract]
Latest Updates to This Summary (04/18/2025)
The PDQ cancer information summaries are reviewed regularly and updated as new information becomes available. This section describes the latest changes made to this summary as of the date above.
Updated statistics with estimated new cases and deaths for 2025 (cited American Cancer Society as reference 1).
This summary is written and maintained by the PDQ Screening and Prevention Editorial Board, which is editorially independent of NCI. The summary reflects an independent review of the literature and does not represent a policy statement of NCI or NIH. More information about summary policies and the role of the PDQ Editorial Boards in maintaining the PDQ summaries can be found on the About This PDQ Summary and PDQ® Cancer Information for Health Professionals pages.
About This PDQ Summary
Purpose of This Summary
This PDQ cancer information summary for health professionals provides comprehensive, peer-reviewed, evidence-based information about cervical cancer prevention. It is intended as a resource to inform and assist clinicians in the care of their patients. It does not provide formal guidelines or recommendations for making health care decisions.
Reviewers and Updates
This summary is reviewed regularly and updated as necessary by the PDQ Screening and Prevention Editorial Board, which is editorially independent of the National Cancer Institute (NCI). The summary reflects an independent review of the literature and does not represent a policy statement of NCI or the National Institutes of Health (NIH).
Board members review recently published articles each month to determine whether an article should:
- be discussed at a meeting,
- be cited with text, or
- replace or update an existing article that is already cited.
Changes to the summaries are made through a consensus process in which Board members evaluate the strength of the evidence in the published articles and determine how the article should be included in the summary.
Any comments or questions about the summary content should be submitted to Cancer.gov through the NCI website’s Email Us. Do not contact the individual Board Members with questions or comments about the summaries. Board members will not respond to individual inquiries.
Levels of Evidence
Some of the reference citations in this summary are accompanied by a level-of-evidence designation. These designations are intended to help readers assess the strength of the evidence supporting the use of specific interventions or approaches. The PDQ Screening and Prevention Editorial Board uses a formal evidence ranking system in developing its level-of-evidence designations.
Permission to Use This Summary
PDQ is a registered trademark. Although the content of PDQ documents can be used freely as text, it cannot be identified as an NCI PDQ cancer information summary unless it is presented in its entirety and is regularly updated. However, an author would be permitted to write a sentence such as “NCI’s PDQ cancer information summary about breast cancer prevention states the risks succinctly: [include excerpt from the summary].”
The preferred citation for this PDQ summary is:
PDQ® Screening and Prevention Editorial Board. PDQ Cervical Cancer Prevention. Bethesda, MD: National Cancer Institute. Updated <MM/DD/YYYY>. Available at: /types/cervical/hp/cervical-prevention-pdq. Accessed <MM/DD/YYYY>. [PMID: 26389433]
Images in this summary are used with permission of the author(s), artist, and/or publisher for use within the PDQ summaries only. Permission to use images outside the context of PDQ information must be obtained from the owner(s) and cannot be granted by the National Cancer Institute. Information about using the illustrations in this summary, along with many other cancer-related images, is available in Visuals Online, a collection of over 2,000 scientific images.
Disclaimer
The information in these summaries should not be used as a basis for insurance reimbursement determinations. More information on insurance coverage is available on Cancer.gov on the Managing Cancer Care page.
Contact Us
More information about contacting us or receiving help with the Cancer.gov website can be found on our Contact Us for Help page. Questions can also be submitted to Cancer.gov through the website’s Email Us.